What Is Deep Brain Stimulation For Parkinsons Disease
Deep brain stimulation is a surgical procedure used to treat a variety of disabling neurological symptomsmost commonly the debilitating symptoms of Parkinsons disease , such as tremor, rigidity, stiffness, slowed movement, and walking problems. The procedure is also used to treat essential tremor, a common neurological movement disorder. At present, the procedure is used only for patients whose symptoms cannot be adequately controlled with medications.
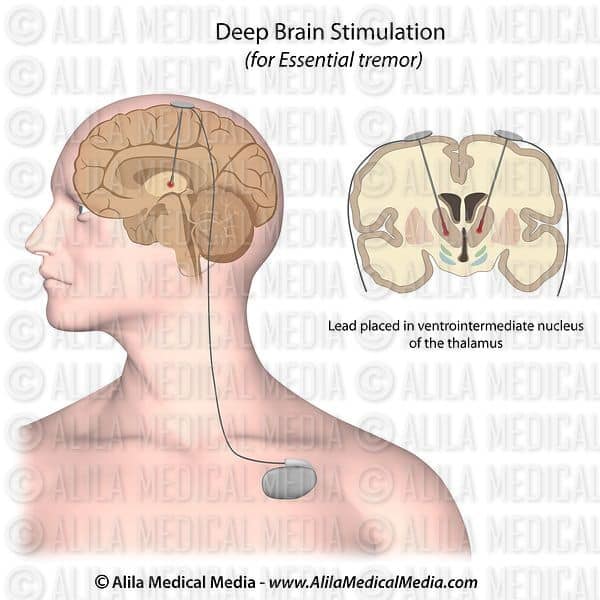
DBS uses a surgically implanted, battery-operated medical device called a neurostimulatorsimilar to a heart pacemaker and approximately the size of a stopwatchto deliver electrical stimulation to targeted areas in the brain that control movement, blocking the abnormal nerve signals that cause tremor and PD symptoms.Before the procedure, a neurosurgeon uses magnetic resonance imaging or computed tomography scanning to identify and locate the exact target within the brain where electrical nerve signals generate the PD symptoms.
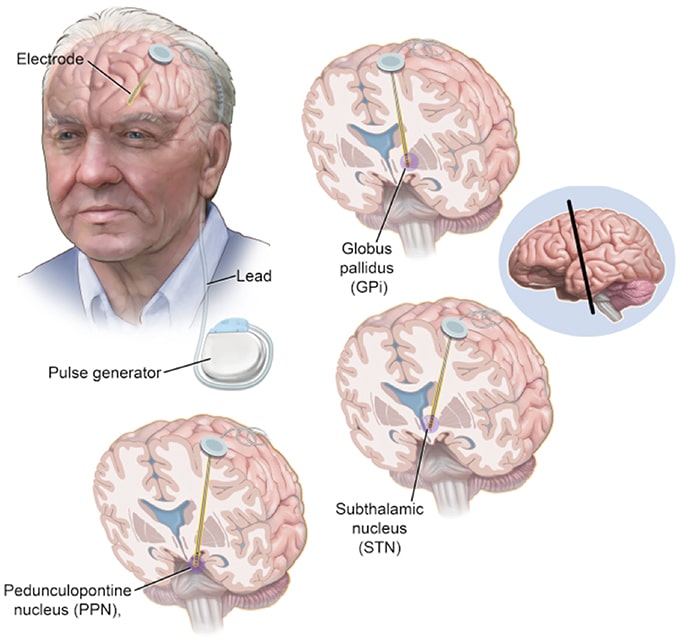
Some surgeons may use microelectrode recordingwhich involves a small wire that monitors the activity of nerve cells in the target areato more specifically identify the precise brain target that will be stimulated. Generally, these targets are the thalamus, subthalamic nucleus, and globus pallidus.The DBS system consists of three components: the lead, the extension, and the neurostimulator.
What Is The Prognosis
Although most people still need to take medication after undergoing DBS, many people experience considerable reduction of their PD symptoms and can greatly reduce their medications. The amount of reduction varies from person to person. The reduction in dose of medication can lead to decreased risk of side effects such as dyskinesia.
There is a 1 to 3% chance of infection, stroke, bleeding in the brain, or other complications associated with anesthesia. It is best to discuss associated risks with your neurologist and neurosurgeon, as diabetes and heart and lung conditions all may influence these risks and the decision to pursue surgery.
Deep Brain Stimulation Care For Movement Disorders
At the USC Deep Brain Stimulation Center, we provide care to decrease the symptoms of dystonia, essential tremor and other movement disorders, such as Parkinsons disease. We use deep brain stimulation when other medical and psychological treatments do not improve your condition.
Deep brain stimulation acts like a pacemaker for your brain. The device blocks abnormal electrical signals in your brain that can cause uncontrolled movements. It can also improve mobility for people who are slowed down by Parkinsons disease. It doesnt cure movement disorders, but it does relieve symptoms so you can be more independent and comfortable.
Don’t Miss: How Many Forms Of Parkinson’s Are There
Research To Improve Deep Brain Stimulation
Researchers are working to improve upon existing DBS devices and methods to help treat more symptoms and more people. Some researchers are putting electrodes in a different area of the brain the pedunculopontine nucleus to treat walking and balance problems that don’t typically improve with present-day DBS. Others are developing a “smart” DBS device that can record a person’s unique brain signals and deliver electrical stimulation only when needed, such as when symptoms return, rather than continuously, as the current systems do. This could help reduce side effects such as numbness and weakness and lengthen the battery life of the neurostimulator, which would result in a longer time between battery replacement procedures.
Scientists also are planning to test deep brain stimulation in the first years after a Parkinson’s diagnosis to see if the therapy may slow or stop disease progression. Testing in Parkinson’s models showed the therapy may help protect brain cells, and a small human trial showed motor symptoms improved after early-stage DBS.
Guidelines With Unclear Methodology
- AndersonDG, Van CollerR, CarrJ. South African guideline on deep brain stimulation for Parkinsons disease. S Afr Med J. 2017 107:10271032.
- FoxSH, KatzenschlagerR, LimSY, et al. International Parkinson and movement disorder society evidence-based medicine review: update on treatments for the motor symptoms of Parkinsons disease. Mov Disord. 2018 33:12481266.
You May Like: Can An Emg Diagnose Parkinson’s
Why Is Dbs Used
There are billions of neurons in each human brain, and these cells communicate with each other using electrical and chemical signals. Several brain conditions can make neurons in different parts of your brain less active. When that happens, those parts of your brain dont work as well. Depending on the part of the brain affected, you can have disruptions in the abilities controlled in that area.
DBS uses an artificial electrical current to make those neurons more active, which can help with the symptoms of several different brain conditions. However, researchers still dont know exactly how or why this works.
Summary Of Study Characteristics
Additional details regarding the strengths and limitations of included publications are provided in .
Systematic Reviews
Clinical Studies
Economic Evaluations
Guidelines
The included guideline met the majority of required criteria of the AGREE II tool. Strengths of the guidelines are the overall objectives and populations to whom the guidelines apply are specifically described guideline development groups included individuals from all relevant professional groups the target users of the guidelines are clearly defined systematic methods were used to search for evidence the criteria for selecting the evidence, the strengths and limitations of the body of evidence, and the methods for formulating the recommendations are clearly described the health benefits, side effects, and risks have been considered in formulating the recommendations there is an explicit link between the recommendations and the supporting evidence the guideline was externally reviewed by experts prior to its publication a procedure for updating the guideline is provided the recommendations are specific and unambiguous and the different options for management of the condition or health issue are clearly presented. These features may increase the reliability of the recommendations as they demonstrate sound methodology and make these publications less prone to biases.
Read Also: Effects Of Parkinson’s Disease On The Elderly
Treatment For Parkinson’s Disease
While there is currently no cure, there are many available treatments that can improve quality of life, sometimes to the point where the disease has little impact. In the earliest stages of Parkinson’s, there may be no reason for treatment. However, if symptoms result in a functional disability that interferes with everyday activities, then medication may be considered.
Sometimes, the medications stop providing significant relief of symptoms. If this occurs, surgical interventions in the basal ganglia may be used to alleviate some symptoms, particularly tremor, rigidity, and dystonic muscle pain and, to a lesser extent, hypokinesia. The procedures performed involve the implantation of deep brain stimulators. Surgery for Parkinson’s disease is performed stereotactically. This means that, under local anesthesia, the targeted areas of the brain are reached with the aid of a computerized guidance system through a small hole in the skull. This type of surgery takes less than two hours and the patient can usually go home the next day.
His Right Leg Became Less Dyskinetic
Another positive change that Dad experienced after DBS was less overall dyskinesia, and especially in his right leg: When I used to come off of levodopa, my right leg would shake uncontrollably for 10 to 15 minutes, and thats gone away, he said. DBS totally took dyskinesia out of my right leg.
Since dyskinesia was one of the main reasons Dad decided to undergo DBS, I was happy to learn that it tackled some of his worst symptoms.
While Dad was initially worried about the potential negative side effects of undergoing DBS, hes a believer in the procedure now. I think the DBS works, and it continues, he said. It improves a little at a time. Its been four years, and it still functions quite well.
As for what Ive witnessed, I think Dad is experiencing a slower progression than he was before DBS. And for that, Ill be eternally grateful to his surgeon.
Also Check: Does Neil Diamond Have Parkinsons
When Should I Call My Healthcare Provider Or Go To The Hospital
Because DBS involves surgery especially the procedure on your brain there are some warning signs you shouldn’t ignore. You should call your healthcare provider immediately or go to the hospital outside of business hours if you have the following symptoms:
- Severe headache that happens suddenly or wont go away.
- Bleeding from your incisions.
A Systematic Review And Guideline Comparing Subthalamic Nucleus Deep Brain Stimulation To Globus Pallidus Internus Deep Brain Stimulation For The Treatment Of Patients With Pd
- RughaniA, SchwalbJM, SidiropoulosC, et al. Congress of Neurological Surgeons systematic review and evidence-based guideline on subthalamic nucleus and globus pallidus internus deep brain stimulation for the treatment of patients with Parkinsons disease: executive summary. Neurosurgery. 2018 82:753756..
Also Check: Alternative Treatment For Parkinson’s Disease
Correlation Of Beta Power And Motor Impairment
Fig. 1: Low beta power in the OFF-medication state and its reduction with dopamine correlate with symptom severity.
A Averaged power spectra across contact pairs in the ON and OFF medication state. Note the decrease in relative power with dopaminergic medication that includes the low beta and sub-beta band . B Matching the frequency bins that show a significant modulation with dopamine, the amplitude of frequencies between 9 and 22Hz show a significant correlation with motor symptoms as assessed by the UPDRS-III. However, symptom alleviation with dopamine is best reflected by amplitude changes from 13 to 19Hz, commonly referred to as the low beta band. In blue are shown Rho-values for each frequency bin, the scattered gray line shows the correspondent P-value, and significant areas are underlined in gray. C There is a significant correlation between symptom severity and averaged low beta power in the dopamine-depleted state . Likewise, the reduction in low beta power with dopamine correlates with symptom alleviation .
Surgical Procedure And Clinical Results
The intraoperative results of microrecording and microstimulation were used to direct macroelectrode implantation into one of the five tracks investigated. The central track was chosen more often than the anterior , medial , lateral , or posterior tracks .
Table 1
Tracks chosen for permanent electrode implantation
Following implantation of the quadrupolar electrode , contacts resulting in the best alleviation of rigidity with the lowest voltage and without side effects were considered optimal and selected for permanent stimulation. As shown in table 2, contact 2 was chosen most often for permanent stimulation. Contacts 1 and 3 were also often selected, whereas contact 0 was rarely chosen . Only one of 25 patients required unilateral stimulation in a bipolar mode.
Table 2
Electrode contacts chosen for permanent stimulation
Figure 1
Unified Parkinsons disease rating scale motor score determined preoperatively and three and 12 months after surgery. MED, suprathreshold dose of levodopa DBS, stimulation with optimal contacts and stimulation parameters.
Also Check: What Are The Non Motor Symptoms Of Parkinson’s Disease
Pallidal Stimulation Versus Subthalamic Stimulation
As mentioned, there are two main anatomic targets for using DBS to treat PD the STN and the GPi. There have been several large randomized studies comparing STN and GPi DBS in PD. It is suggested that both STN DBS and GPi DBS overall equally and successfully improve motor symptom, and are similar in cost-effectiveness. However, although no differences were observed in the on phase between STN DBS and GPi DBS, significant differences were seen in the off phase STN DBS was more effective in terms of motor function improvement in the off phase. There are different opinions in terms of effects of STN DBS and GPi DBS on quality of life. Some authors have found no significant difference between the STN and GPi targets. However, others agree with that greater improvements in quality-of-life measures are achieved in patients with GPi DBS.
GPi DBS can be used for patients with more axial symptoms, gait issues, dyskinesias, depression, and word fluency problems. STN DBS is often favored in reducing medication post surgery, and for patients with greater tremor. STN-DBS has also demonstrated an improvement in the quality of sleep for patients.
Stereotactic Dbs Vs Interventional Image
Stereotactic DBS surgery requires the patient to be off their medication. During the procedure, a frame stabilizes the head and provides coordinates to help the surgeons guide the lead to the correct location in the brain. The patient gets local anesthesia to keep them comfortable throughout each step along with a mild sedative to help them relax.
During image-guided DBS surgery, such as with interventional MRI or CT scan, the patient is often asleep under general anesthesia while the surgeon uses images of the brain to guide the lead to its target.
Some advanced centers offer both the stereotactic and iMRI-guided options for DBS surgery. In this case, the doctor and patient will discuss which procedure is better based on a number of factors.
For instance, the doctor may recommend an image-guided procedure for children, patients who have extreme symptoms, those who are especially anxious or fearful or those whose leads are going into certain parts of the brain.
Generally, DBS surgery follows this process:
Don’t Miss: Is Head Shaking A Sign Of Parkinson’s
Less Medication More Relief
Medtronic DBS therapy may reduce the need for other Parkinsons medications1 and, consequently, medication-related side effects. DBS delivers therapy 24 hours a day and doesnt wear off while sleeping. Its already working when you wake up.
* Signal may not be present or measurable in all patients. Clinical benefits of brain sensing have not been established.
Coordinates Of Active Electrode Contacts
Retrospective analysis of the stereotactic position of the active electrode contacts was done in 25 patients for whom postoperative T1 weighted MRI of sufficient quality or stereotactic radiographic examinations were available. Such analyses could not be undertaken in other patients implanted with subthalamic nucleus electrodes during the same period because of motion artefacts in the postoperative MRI, missing postoperative T1 weighted MRI, or missing postoperative stereotactic x rays. For the 25 patients evaluated, the mean and median coordinates of all active contacts are summarised in table 3. The mean laterality of all active electrode contacts mm median 12.7 mm) correlated well with the laterality of the subthalamic nucleus, as determined 3 mm ventral to the intercommissural plane in T2 weighted MRI of 35 patients mm). However, in the dorso-ventral direction the mean and median z coordinate of all active contacts do not project within the subthalamic nucleus proper, but suggest an area between the dorsal margin of the subthalamic nucleus and the subthalamic region according to different stereotactic brain atlases. Moreover, 12 of 49 active contacts were located within 0.5 mm of the intercommissural plane or further dorsal they were thus most probably in the subthalamic area.
Active electrode contacts relative to the mid-commissural point
Don’t Miss: Is Gabapentin Used For Parkinson’s Disease
Critical Appraisal Of Individual Studies
The included systematic reviews were critically appraised by one reviewer using A MeaSurement Tool to Assess systematic Reviews 2, randomized studies were critically appraised using Downs and Black checklist, economic studies were assessed using the Drummond Checklist, and guidelines were assessed with the Appraisal of Guidelines, Research and Evaluation II instrument. Summary scores were not calculated for the included studies rather, a review of the strengths and limitations of each included study were described narratively.
Dbs Becomes A Clinical Success
During a series of surgeries to treat patients with PD and other motor disorders who were not helped by L-DOPA, French neurosurgeon Alim-Louis Benabid observed that applying high frequency stimulation to the ventral intermediate nucleus of the thalamus caused the patients tremor to cease23. Additional small trials24,25 and Medtronic-funded multisite trials in the US and Europe confirmed the treatments effectiveness and showed that thalamic stimulation was safer than surgical inactivation. In 1997, the FDA granted approval for Medtronics device for VIM-DBS to treat essential tremor and tremor associated with PD26.
Subsequent studies have continued to assess long term outcomes of DBS, optimize targets for different symptom profiles, and compare the effectiveness of DBS with medical treatment. In 2009, a pivotal clinical trial jointly supported by NINDS, the Department of Veterans Affairs, and Medtronic demonstrated that DBS for PD was superior to treatment with L-DOPA33. With growing evidence for the safety of DBS and results suggesting earlier intervention in PD may be beneficial, the FDA expanded approval beyond advanced stages of PD in 2016, to allow treatment in patients diagnosed for at least four years who experience troublesome off periods or dyskinesia34.
Don’t Miss: Living With Parkinson’s Disease Video
How Does Deep Brain Stimulation For Parkinsons Work
Deep brain stimulation works by modifying abnormal electrical activity in the brain. It was first approved for Parkinsons tremors in 1997 and has become an established treatment to control additional motor symptoms of Parkinsons disease.
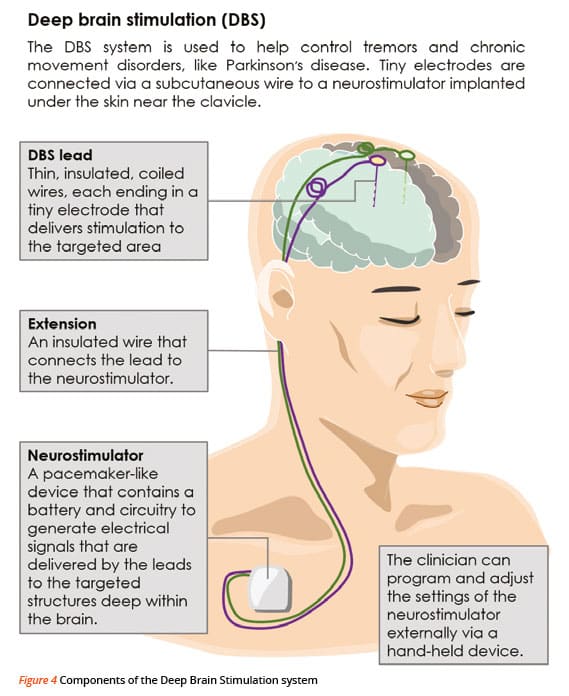
DBS involves three main components:
- Leads: Leads are implanted in the brain in a region responsible for motor activity.
- Implantable pulse generator : A separate procedure is performed to implant a battery-operated device in the chest or in the abdomen. An IPG is similar to a pacemaker for the heart and has been coined by some as a pacemaker for the brain.
- Extension: A thin, insulated wire is passed beneath the skin between the leads and implantable pulse generator to deliver the electrical stimulation from the pulse generator to the leads.
The target area in the brain is first identified by magnetic resonance imaging or computed tomography . Then, the leads are placed via small holes that a surgeon drills in the skull.
This is considered a minimally invasive surgery that is done in the operating room with local anesthesia. It usually requires an overnight stay.
The IPG is inserted in a separate surgical procedure in the operating room roughly a week later.
After a few weeks, a neurologist begins to program the unit. This process can take several additional weeks to months. When this is completed, people are able to manage the device with a handheld remote control.
Deep Brain Stimulation At Michigan Medicine
For carefully selected patients with Parkinsons disease, Essential Tremor, and Dystonia, deep brain stimulation offers a therapeutic surgical option that can reduce or eliminate movement-related problems and greatly improve quality of life. At the University of Michigan Health System, our STIM program brings together a team of medical experts who are leaders in their respective fields and on the cutting-edge of the latest research.
Recommended Reading: Do Parkinson’s Symptoms Come And Go
