How Are We Using Stem Cells To Understand And Treat Parkinsons Disease
Stem cells are being used in multiple ways to understand the disease process and find new ways to potentially treat PD. By using stem cells in the lab to either model the disease by studying the affected neurons under the microscope or make healthy neurons to transplant into patients to replace the cells lost to the disease, stem cells are helping scientists discover new clues about how the disease is triggered and how it might one day be treated.
Etiology And Risk Factors
Parkinsonian symptoms can arise from either the neuropathologic condition of PD or other forms of parkinsonism. For neuropathologic PD, about 90% of cases are sporadic, with no clear etiology an additional 10% have a genetic origin, and at least 11 different linkages with 6 gene mutations have been identified Genetic forms of PD are seen more frequently in young-onset PD. A combination of environmental factors or toxins, genetic susceptibility, and the aging process may account for many sporadic cases Secondary forms of parkinsonism can be caused by medications, the sequelae of central nervous system infection, toxins, or vascular/metabolic disorders . The only proven risk factor for PD is advancing age . Other environmental or lifestyle risk factors associated with development of PD are rural living, exposure to pesticides and herbicides, well-water drinking, and working with solvents . However, none of these factors unequivocally has been demonstrated to cause iPD .
Figure 1: Etiology of Parkinson’s Disease . View Figure 1
Is It Safe Safety Of Commercial Stem Cell Clinic Work
Safety data is also limited, although there have been some publicized lawsuits claiming that these treatments resulted in harm. Stem cell researchers in general question whether cells harvested in such a way contain sufficient amounts of adult-derived stem cells to be meaningful. It is also unclear how this type of procedure would target the stem cells to the correct location. If stem cells are introduced in the nose for example, it is unclear how they would find their way to the basal ganglia and make the correct connection in order to help a person with Parkinsons disease.
In order for the medical community to accept this type of treatment as safe and beneficial, it would need to be shown to work in a placebo-controlled clinical trial for which participants do not pay, are aware of the known risks and benefits, and are carefully monitored throughout the trial. In addition, the trial would need to track adverse events, as well as record and share the outcomes of trial participants as they compare to the group of patients receiving a placebo treatment. So far this has not happened. The FDA is in fact studying mesenchymal stem cells in the laboratory in order to determine the best way to use them to help people, but these studies have not yet led to approved treatments. Most recently, the FDA filed federal complaints against two clinics that are marketing stem cell products without regulatory approval.
You May Like: How To Get Tested For Parkinson’s Disease
What Are Stem Cell Lines
A stem cell line is a family of constantly dividing cells, the product of a single parent group of stem cells. They are obtained from human or animal tissues and have been manipulated in a laboratory so that they have the ability to divide almost indefinitely, creating the line.
Because stem cell lines produce so many copies of themselves , this means that scientists have a large bank of cells for their research and are less likely to need to take cells from an embryo repeatedly. Of course, it is important that researchers are also able to stop such cell lines dividing at some point so that they can generate tissue specific cells, such as for the brain in the case of Parkinsons treatment. The challenge for researchers is to discover how they can control the process which causes stem cells to differentiate.
Induced Pluripotent Stem Cells
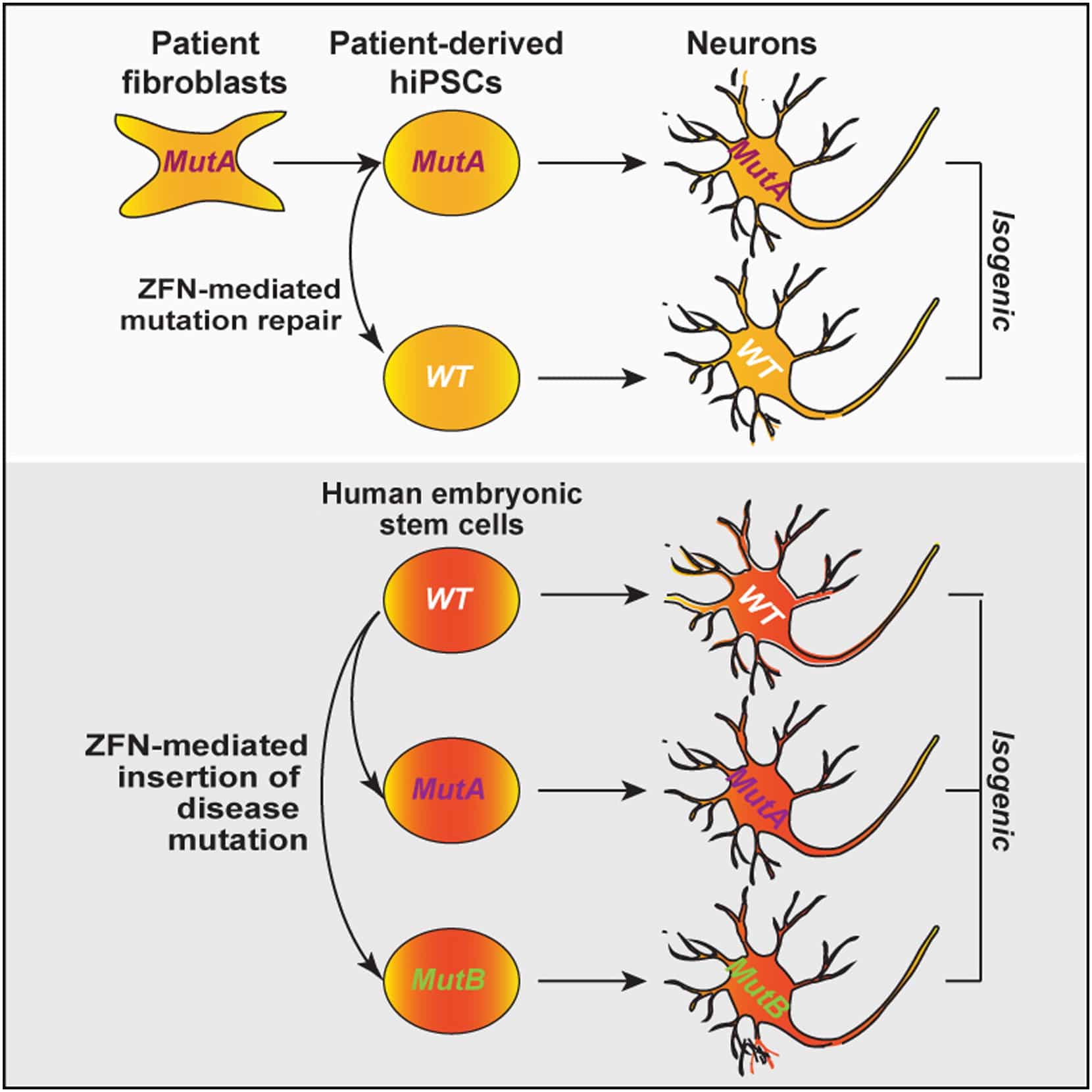
In 2007, the process for generating iPSCs was first reported, offering a new avenue for the development of a stem cell-based treatment for PD . iPSCs are generated by the reprogramming of an adult somatic cell into a stem cell, through the expression of a number of transcription factors that could induce pluripotency . The iPSCs derived in this way can be differentiated into dopaminergic neurons using protocols similar to those used with ESCs, which could serve as the basis of a useful cell-based treatment for PD . The potential advantage of iPSC-derived over ESC-derived grafts is that it would be possible to generate autologous grafts, by using a patients own fibroblasts to produce a neural grafting product, negating the requirement for immunosuppression that will be necessary with ESC-derived grafts. However, there are other biological and logistical challenges faced with the iPSC approach, which are discussed below.
iPSC-derived neural grafts have been trialed in primates with MPTP-induced nigral toxicity, with promising results . The neural progenitors grafted ultimately extended neurites into the striatum, did not form any tumors, and resulted in improved motor function at two years. As with the ESC-approach, clinical trials in humans are on the horizon and will begin in the next couple of years .
Also Check: A Typical Parkinson’s Progression
How Are Stem Cells From Stem Cell Supplement Administered For Parkinsons Disease
Unlike traditional Stem Cell Therapy which can only be applied via injection in the selective hospitals/ clinics, Stem Cell Supplement delivers stem cells through high-tech bio-active softgel capsules which can be shipped in secure packages and delivered right to your home. The lyophilized method employed in Able softgel capsules produces stem cells which remain biologically active without damaging the effectiveness of the valuable, big, bio-active matter. Being enteric coated, Able softgel capsules by-pass the stomach and dissolve in the small intestine whereby the stem cells and other active ingredients are fully absorbed by the body. Therefore, to receive the results and benefits of Stem Cell Supplement, all you need to do is to take 1 3 softgel capsules a day at the comfort of your own home.
Also Check: Parkinsons Disease Medications Carbidopa Levodopa
Mesenchymal Stem Cell Therapies For Neurodegenerative Diseases
While there have been significant advances in the symptomatic management of these diseases that improve quality of life and at times survival, the available medications likely only slow the progression of neuronal death by a few months. The idea of using cell therapy to treat neurodegenerative diseases has been around for decades, most notably in Parkinson’s Disease where a variety of cell transplant investigations have been performed with success.
According to a recent study conducted by Nathan P. Staff et al,
“The precise mechanism by which MSCs may exert beneficial effects in neurological disease is still being elucidated, but it appears that multiple different mechanisms may contribute. First, MSCs have been shown to secrete neurotrophic growth factors, including glial cell-derived neurotrophic factor , vascular endothelial growth factor, and brain-derived neurotrophic factor ,which can be further enhanced under specific culture conditions.Neurotrophic growth factors have been shown to improve neuronal survival in a number of preclinical models of neuron injury, including ALS, PD, and MSA transgenic animalsand nerve injury models. â Second, MSCs strongly modulate the immune system and can aid wound healing, and this mechanism has been exploited in disorders such as graft versus host disease and Crohnâs disease. From a neurodegenerative perspective, it has become increasingly recognized that neuroinflammation plays a significant pathomechanistic role.”
Recommended Reading: What Are The Different Types Of Parkinson’s Disease
Mda Cell Dosage For Transplantation
Typically, 200,000420,000 dopamine neurons reside in human midbrain, and it is estimated that 50% loss of those DA neurons leads to the PD symptom . According to preclinical studies using fetal tissue or hPSC-derived mDA cell, 12002400 surviving TH + neurons in rat, 13,000 in primate, 40,00080,000 in the human brain may be required to achieve a meaningful therapeutic effect . The current bottleneck in delivering cells to the brain is that typically less than 10% of grafted mDA neurons survive following transplantation . Multiple factor may contribute to poor mDA neuron survival including mechanical trauma, growth factor deprivation, initial lack of vascularization, hypoxia and free radical production, or excessive extracellular concentrations of excitatory amino acids in the host brain .
Pure Substantia Nigra Mda Neuron
Recently, single cell gene profiling has been used to define subtype compositions during mouse and human midbrain development . Such technology extended the previous molecular definition of mDA neurons and enabled to further divide SN and VTA region into seven distinct molecular clusters . However, to what extent these molecular findings can be translated into hPSC-derived mDA neuron development remains unexplored. This is illustrated when profiling of hPSC-derived mDA neurons in La Manno et al., which seem to recapitulate key stages of in vivo ventral midbrain development. However, those cell preparations expressed many poorly defined radial glial and neuroblast markers and differed from the in vivo phenotypes in gene expression . Additionally, ALDH1A1 was not expressed in any of those PSC-derived mDA cells. This result may be due to the in vitro culture environment that does not fully support mDA neuron development or lack of proper induction of certain subtype-specific genes. In either case, those results indicate that there is considerable room for further improvements in mDA neuron derivation and maturation strategies.
Don’t Miss: Nutrition For Parkinson’s Disease
The Next Generation Of Trials
Studer was part of the initial studies involving fetal tissue in the 1980s and 1990s, and knew from the start that the work was more of a proof of principle than a solution for people with Parkinsons. For me it was clear that a fetal transplant isnt a long-term solution because of ethical, legal and practical issues. Because this procedure requires 4 to 12 fetuses per patient, there was no way they could treat thousands, let alone tens of thousands, of people that way. Instead, Studer turned to stem cells.
Immunosuppression is a particularly important element of BlueRocks approach, because it relies on a single cell line that cannot be adjusted to more closely resemble the recipients own tissues. A group led by stem-cell scientist and neurosurgeon Jun Takahashi at Kyoto University in Japan is attempting to provoke a lesser immune response by pairing transplant recipients with cells that are less likely to be rejected. The researchers are using cell-surface proteins, called major histocompatibility complexes , that are recognized by the adaptive immune system and can have varying levels of compatibility from one person to another. Rather than using frozen cell lines, Takahashi and his colleagues are creating a fresh batch of MHC-matched cells for each transplant.
Does It Work Efficacy Of Commercial Stem Cell Clinics
Commercial clinics do not as a rule publish their results in peer-reviewed journals to demonstrate to the scientific community that the treatments work. Rather, they usually rely on anecdotes from patients as proof of efficacy. Some clinics are tracking their results by measuring variables such as quality of life before or after the procedure. However, without comparing the patients to a similar group who does not receive the treatment, it is hard to know whether any improvement is due to placebo effect or to the treatment itself.
Read Also: Is Parkinson’s A Genetic Disorder
What Are Human Embryonic Stem Cells And How Are They Obtained
Human embryonic stem cells are the cells from which all 200+ kinds of tissue in the human body originate. Typically, they are derived from human embryosoften those from fertility clinics who are left over from assisted reproduction attempts . When stem cells are obtained from living human embryos, the harvesting of such cells necessitates destruction of the embryos.
Stem Cell Therapy For Parkinson’s Disease
Stem cell therapy may have the benefit of replacing and repairing damaged dopamine-producing nerve cells within the brain. This has already been found in a study conducted by Neelam K.Venkataramana and colleagues. Seven PD patients aged 22 to 62 years with a mean duration of disease 14.7 ± 7.56 years were enrolled to participate in the prospective, uncontrolled, pilot study of single-dose, unilateral transplantation of autologous bone-marrow-derived mesenchymal stem cells . Patients were followed up for 36 months post-transplant, 3 of the 7 patients showed significant improvement in their Unified Parkinson’s Disease Rating Scale of 38%.
According to Medical News Today “Currently, the most common therapy uses the drug levodopa to stimulate dopamine production in certain neurons associated with motor skills. These dopaminergic neurons are situated in the nigrostriatal pathway which is a brain circuit that connects neurons in the substantia nigra pars compacta with the dorsal striatum. However, levodopa has a wide array of side effects, from physiological to psychological ones. Also, in the long-term, the benefits of such dopamine-regulating drugs are limited. So, scientists must come up with more effective strategies for repairing the brain damage that Parkinson’s disease causes.”
You May Like: What Are Risk Factors For Parkinson’s Disease
Have Scientists Been Successful In Using Embryonic Stem Cells To Treat Disease
Though embryonic stem cells have been purported as holding great medical promise, reports of actual clinical success have been few. Instead, scientists conducting research on embryonic stem cells have encountered significant obstaclesincluding tumor formation, unstable gene expression, and an inability to stimulate the cells to form the desired type of tissue. It may indeed be telling that some biotechnology companies have chosen not to invest financially in embryonic stem cell research and some scientists have elected to focus their research exclusively on non-embryonic stem cell research.
Induced Pluripotent Cells Are Not Surrounded By The Same Moral Issues As Hesc
In parallel with the development of medical therapy using hESC, researchers have succeeded in producing induced pluripotent stem cells from human cells and using them for medical product development . Unlike hESC, iPS-cells are derived from non-germ cells and thus are not surrounded by the same moral concerns Researchers are currently developing medical products to replace damaged or dead cells for various chronic diseases, e.g., for Parkinsons disease . Currently, the aetiology of PD is still unknown. There are no disease modifying therapies available for patients rather, therapy focuses on symptom relief by compensating for low dopamine levels in the brain. Commonly, patients daily lives are increasingly affected over time by symptoms such as tremors, slow movements and balance problems. It is common to develop non-motor problems like depressive symptoms and, later, dementia. As the symptoms get worse with time, medicines are often given more frequently, and device-aided therapies are introduced. It is not uncommon for patients to suffer from side-effects of treatment, such as dyskinesia or behavioural problems.
Oncogenic mutations have been reported in both transplant types. However, there is still debate on whether stem cell therapy is feasible, efficient and safe, and whether it affects enough of the PD neuropathology. Several methods have been developed to prevent tumour formation .
Recommended Reading: Is Difficulty Swallowing A Symptom Of Parkinson Disease
Stem Cells For Parkinsons: Therapy And Tools For A Neurological Disorder
This is a guest post from The Michael J. Fox Foundation for Parkinsons Research . MJFF is committed to the pursuit of a Parkinsons cure and better quality of life for those living with the disease today. Stem cells are valuable tools in that work, helping develop new therapies and learn more about the disease. Find out more about the work they do at www.michaeljfox.org.
Parkinson’s disease is a neurological disorder that affects one in 100 people over age 60. The disease causes a variety of symptoms including motor problems such as tremors, muscle rigidity and slowed movement, and non-motor symptoms of cognitive impairment, mood disorders, and autonomic dysfunction. It is estimated that nearly 1 million people in the United States and more than 6 million worldwide have Parkinsons disease. Current treatments can ease some symptoms, but no available therapies stop or slow the progression of the disease.
Scientists are using stem cells to better understand and treat Parkinsons disease.
Stem Cell TreatmentsIn Parkinsons disease , cells that make the chemical messenger dopamine degenerate and die. Introducing new dopamine cells into the brain may help replace what is lost in PD and reduce its symptoms. Such a treatment also could reduce medication side effects. Long-term use of the most commonly prescribed PD medication and progressing disease can lead to dyskinesia or uncontrolled, involuntary movements.
Gmp Cryopreservation Of Cells
The generation of good manufacturing practice -compliant, deliverable midbrain DA progenitors/neurons optimized for cell-based therapy for PD is a major challenge. Currently, a diverse collection of clinical-grade hESC lines are available as starting material to generate GMP-compliant mDA progenitors/neurons. In fact, GMP compliant differentiation protocols and reagents have been successfully applied to generate GMP mDA neurons .
Recommended Reading: Does Black Seed Oil Help Parkinson’s Disease
Studies Show Promising Results
“Considering the ability of MSCs to secrete neurotrophic factors, modulate inflammation, and possibly even act as mitochondria âdonorâ, it comes as no surprise that there is a lot of interest in the use of MSCs in the treatment of Parkinsons Disease, and a multitude of animal studies has shown promise. Treatments have resulted in improvement of motor function, protection of the nigrostriatal system, and improved striatal dopamine release in several studies using toxic lesion rodent models of Parkinsons Disease. Similar effects were reported with umbilical cord-derived MSCs with or without prior differentiation. For example, a recent study reported improvement of motor function, reduced microglial activation, and decreased loss of TH immunoreactivity, associated with local production of trophic factors.
Learn more about DVC Stem’s protocol for Parkinson’s Disease here:
References:
Venkataramana, N. K., Kumar, S. K. V., Balaraju, S., Radhakrishnan, R. C., Bansal, A., Dixit, A., ⦠Totey, S. M. . Open-labeled study of unilateral autologous bone-marrow-derived mesenchymal stem cell transplantation in Parkinson’s disease. Retrieved from https://www.sciencedirect.com/science/article/pii/S1931524409002205#!
Unified Parkinson’s Disease Rating Scale. . Retrieved from https://www.sciencedirect.com/topics/medicine-and-dentistry/unified-parkinsons-disease-rating-scale
About the author
