Are Alterations In The Axonal And Synaptic Mitochondrial Populations Correlated With Respiratory Deficiency In The Soma
Although in human tissue it is not possible to compare cell body changes to synaptic/axonal changes within the same neuron, a measure of respiratory chain deficiency was made within the soma of SN neurons for most of the cases included in this study . Intensity measurements were made for immunoreactivity for NDUFB8, COXI and porin within TH positive cell bodies within the SN. A reduction in expression of either of these proteins was defined as an intensity value that had a z score of less than ?1, normal neurons had a score of between ?1 and 1, while an increase was defined as having a score over 1. These parameters were set based on the control data.
There was an increase in the percentage of cell bodies showing reduced expression of both complex I and complex IV in PD cases compared to other groups, and in DLB cases there was an increase in complex IV deficient neurons, however neither of these changes was significant due to the variability between cases . In addition to measuring deficiency within the neurons of the SN, mitochondrial density was also quantified. Using the signal intensity for porin immunoreactivity we categorised each neuron based on the z score for porin. We again found that there was no significant difference between the mitochondrial mass in cell bodies between any of our patient groups .
Analysis Of The Relationship Between Type Ii Diabetes Mellitus And Parkinsons Disease: A Systematic Review
Fauze Camargo Maluf
1Medical Student of Centro Universitario Saude ABC, Centro Universitario Saude ABC, FMABC, Santo Andre 09060-870, Brazil
2Department of Pharmacology, Centro Universitario Saude ABC, FMABC, Santo Andre 09060-870, Brazil
3Department of Neurosciences, Centro Universitario Saude ABC, FMABC, Santo Andre 09060-870, Brazil
Abstract
1. Introduction
The prevalence of type 2 diabetes mellitus is 370 million people in the world. The T2DM is most frequent in adulthood; however, in the last years, the prevalence of T2DM is increasing in adolescents and children . T2DM is a chronic metabolic disease characterized by long-term insulin resistance and a decrease of ?-cell function and population. These factors impair insulin release and consequently cause hyperglycemia . However, genetic and environmental factors are responsible for 20% of ?-cell failure in the diabetic population .
One of the consequences of chronic diabetes is the production of toxic aggregates of the islet amyloid polypeptide . The IAPP might contribute to ?-cell dysfunction .
Parkinson’s disease affects about 1% of people over 65 and up to 4-5% of people over 85, and thus it represents the second most common neurodegenerative disorder .
The diagnosis of PD is still based on the presence of symptoms and clinical signs such as typical asymmetric manifestation, the most common finding being tremor at rest in the upper limbs associated with bradykinesia, rigidity, and gait difficulty .
3. Results
Breaking Down The Parkinson’s Pathway: How Affected Brain Cells Respond During Different Behavioral Tasks
The key hallmark of Parkinson’s disease is a slowdown of movement caused by a cutoff in the supply of dopamine to the brain region responsible for coordinating movement. While scientists have understood this general process for many years, the exact details of how this happens are still murky.
“We know the neurotransmitter, we know roughly the pathways in the brain that are being affected, but when you come right down to it and ask what exactly is the sequence of events that occurs in the brain, that gets a little tougher,” says Ann Graybiel, an MIT Institute Professor and member of MIT’s McGovern Institute for Brain Research.
A new study from Graybiel’s lab offers insight into some of the precise impairments caused by the loss of dopamine in brain cells affected by Parkinson’s disease. The findings, which appear in the March 12 online edition of the Journal of Neuroscience, could help researchers not only better understand the disease, but also develop more targeted treatments.
Lead author of the paper is Ledia Hernandez, a former MIT postdoc. Other authors are McGovern Institute research scientists Yasuo Kubota and Dan Hu, former MIT graduate student Mark Howe and graduate student Nuné Lemaire.
Cutting off dopamine
As the rats learned to run a T-shaped maze, the researchers recorded electrical activity in many individual neurons. The rats were rewarded for correctly choosing to run left or right as they approached the T, depending on the cue that they heard.
Dorsal Motor Nucleus Of The Vagus Raphe Nuclei Nucleus Basalis Of Meynert And Ventral Tegmental Area
Substantial cell loss has been documented in the DMV, containing cholinergic neurons, with 7 studies evaluating this loss in 49 cases. Of these, only one study used stereology, where they reported 55% neuronal loss in eight PD cases, ranging from 5 to 24 years post-diagnosis and reported correlation with disease duration/severity.
The importance of re-evaluating cell loss is PD is apparent when considering the serotonergic RN. For these nuclei, which are considered by many authors to be lost in PD, we found 7 papers describing neuronal loss varying between 0 to 90%. Cheshire et al. however, using stereology in 44 late-stage PD subjects, found no cell loss in the dorsal raphe nucleus . In the NBM, containing cholinergic neurons, we found 13 papers, 12 using manual counting methods, and one observational, which estimated an average neuronal loss of between “some” to 72%. No correlation with disease duration was reported. The high prevalence of concomitant PD and Alzheimer’s disease might explain why cell loss varied so much for this region. Surprisingly, only 8 studies directly evaluated neuronal loss in the VTA, a dopaminergic region often considered to be only modestly affected in PD. Of these, one study used stereology to evaluate the loss of neurons in 3 cases of PD that were between 1 and 27 years post-diagnosis and reported an average neuronal loss of 31%. One paper reported correlation of the extent of cell loss with disease duration .
Diabetes A Contemporary Risk For Parkinsons Disease: Epidemiological And Cellular Evidences

- 1Nutrition and Health Substantiation Group, Nutrition and Health Program, Health and Biosecurity, Commonwealth Scientific and Industrial Research Organisation , Adelaide, SA, Australia
- 2Adelaide Medical School, University of Adelaide, Adelaide, SA, Australia
- 3Cellular Neurobiology, Department of Medical Biology, Université du Québec, Trois-Rivières, QC, Canada
- 4Department of Biomedical Sciences, University of Cagliari, Cagliari, Italy
- 5National Institute for Neuroscience , University of Cagliari, Cagliari, Italy
- 6Department of Psychiatry and Neuroscience, Université Laval and CHU Research Center, Québec, QC, Canada
Microglial Activation And Systemic Chronic Inflammation Increase The Risk Of T2dm And Pd
Microglia are mononuclear, phagocytic immune cells in the central nervous system. They are normally involved in removal of damaged neurons, and they release neuroprotective factors to promote synaptic regeneration . Microglia can be activated towards either an anti-inflammatory or inflammatory phenotype. For example, microglia can be stimulated by lipopolysaccharide to enter an activated inflammatory state and express pro-inflammatory cytokines such as TNF?, interleukin 1? and IL6, triggering neuroinflammation . A study of 14 patients with PD found evidence of increased microglial activation on PET imaging . Microglial activation is a key contributor to neuroinflammation through the release of inflammatory cytokines , and patients with PD have been shown to have high concentrations of inflammatory mediators such as IL1?, IL6 and TNF? in the brain .
The Relationship Between Synaptic Changes And Cell Body Loss Within The Substantia Nigra
To understand whether the synaptic changes detected were related to the degeneration of SN neurons we quantified the number of cell bodies within the SN and investigated the relationship between synaptic alterations and the amount of cell loss. As expected a significant reduction in cell number was seen in PD and DLB cases , compared to both control and AD cases . Furthermore, we investigated whether any correlation existed between SN cell body loss and either the prevalence of empty dopaminergic synapses within the striatum or with the pre-synaptic volume . No significant relationships were found in either instance. In general, in PD and DLB there was more cell loss seen in cases with fewer empty synapses and this relationship fitted the data most strongly for the PD cases, while a general trend was seen for the pre-synaptic volume to be increased in cases with severe cell loss.
What Should I Do If Im Managing Type 2 Diabetes And Concerned About My Parkinsons Risk
The takeaway of the new analysis for people currently managing or caring for a person with diabetes is unclear. This specific research, for example, doesn’t illustrate how someone with diabetes may help lower their risk of Parkinson’s disease, says Dr. Cereda.
“Unfortunately, although there is some evidence that diabetes is a risk factor for developing Parkinson’s disease, there is no evidence that optimal diabetes control reduces the risk of Parkinson’s disease,” says Cereda.
Yet managing blood sugar is still essential for people with type 2 diabetes, because failing to do this increases the risk of a wide range of health problems including heart disease, stroke, and kidney failure, Cereda says. The study results suggest that we might one day add Parkinson’s disease to the long list of conditions that can be prevented at least in part by good diabetes management, Cereda adds.
Noyce agrees, emphasizing the importance of blood sugar management regardless of potential Parkinson’s risk. “There are many other negative health outcomes that are associated with type 2 diabetes, such as heart disease, stroke, nerve and kidney damage, and visual loss,” Noyce says. “These are all more common than Parkinson’s, and the risk of these things can be reduced with treatment of diabetes, modification of diet, exercise and self-care.”
Type 2 Diabetes Associated With Increased Risk Faster Progression Of Parkinson Disease
A study finds type 2 diabetes associated with an increased risk of developing Parkinson disease , as well as faster progression of motor symptoms in those with PD.
Patients with type 2 diabetes may be at greater risk for developing Parkinson disease , with T2D also associated with faster disease progression in those with PD, according to study findings published this week in Movement Disorders.
As 2 prevalent diseases within an aging population, prior research has highlighted the biologic similarities between T2D and PD.
“Both are characterized by aberrant protein accumulation, lysosomal and mitochondrial dysfunction, and chronic systemic inflammation,” explain the study authors. “Insulin resistance is a hallmark of T2D and may be an important contributing factor to PD, too.”
In addition to studies on the relationship between the 2 diseases, prior systematic reviews and meta-analyses have investigated whether T2D may contribute to risk of developing PD. Although findings are conflicting, the researchers note that most of these studies recruited cohorts of patients with diabetes, as opposed to T2D, with this association also not explored via modern causal methods.
In the meta-analysis, pooled effect estimates showed that T2D was associated with an increased risk of PD , a causal relationship supported by MR .
Reference
Involvement Of Oxidative Stress/mitochondrial Dysfunction In T2dm And Pd Pathogenesis
Mitochondrial proteins, when dysfunctional, produce an increase in oxidative stress and cell death . MPTP exerts its Parkinson’s-like effects in rodent models by selectively inhibiting complex I, the first enzyme in the mitochondrial respiratory chain pathway, leading to neuronal death and neurodegeneration . The features of mitochondrial dysfunction may be shared in T2DM and PD . In PD, dysfunctional insulin signalling has been found to increase oxidative stress , while a recent study showed that chronic insulin resistance in diabetic db/ db mice can cause mitochondrial disruption and dopaminergic neuronal degeneration . Studies using rodent models show that IRS1 and IRS2 inhibit FOXO1 via the PI3K/Akt pathway , resulting in dysfunctional ATP generation and fatty acid oxidation, and the generation of ROS and oxidative stress. While the exact mechanism by which mitochondrial dysfunction and oxidative stress contribute towards PD remains uncertain, its role is likely to be important in PD pathogenesis and potentially relevant to the link with T2DM.
Electrical Activity Dwindles In Cells Long Before Movement Issues Become Visible
- Date:
- University of Texas Health Science Center at San Antonio
- Summary:
- Researchers are studying changes in Parkinson’s-affected cells at various stages of the disease, long before any symptoms are evident. Parkinson’s is marked by the degeneration and death of cells called dopamine neurons. These neurons are found in a brain structure called the substantia nigra. The researchers studied mice in which only these neurons are affected by a genetic mutation.
It’s an unsettling thought: You could be walking around for 20 years developing Parkinson’s disease and not even know it.
And once symptoms appear, it’s too late for a cure.
What if a therapy that treats the root causes of Parkinson’s, not just the symptoms, could be started earlier?
Researchers in the School of Medicine at The University of Texas Health Science Center at San Antonio are studying changes in Parkinson’s-affected cells at various stages of the disease, long before any symptoms are evident. They describe the changes in an April issue of the Journal of Neuroscience.
The hope of the research is twofold: 1) gain understandings that can be used to formulate a drug to arrest the disease at a halfway point, and 2) lengthen the time when patients with Parkinson’s can lead healthy, productive lives.
Hidden changes
Mimics human Parkinson’s
Timeline of decline
With these comparisons, the researchers constructed a timeline of functional decline in the dopamine neurons. They observed changes in three categories:
Study Examines Connection Between Diabetes Medication And Parkinsons Disease
It was first suggested in the 1960’s that people with type-2 diabetes are at increased risk for developing Parkinson’s disease – and when they do develop PD, its progression is faster and often more severe. This may be due, in part, to an apparent relationship in the brain between dopamine, insulin resistance, and glucose control. Insulin is not only made in the pancreas, it’s also present in the brain – where it has been shown to impact dopamine levels.
Parkinson’s is generally believed by scientists to be caused by the loss of dopamine-producing neurons. Parkinson’s symptoms, such as slowness, rigidity, and tremor, typically develop after approximately 40-80% of these dopamine-producing neurons die.
Why does this matter? Currently, more than 30 million people in the United States have type-2 diabetes, and that number is growing. The lifetime risk of developing Parkinson’s is also on the rise. In light of these trends, it would be valuable to know whether any specific type-2 diabetes medications might be associated with an increased or decreased risk for developing PD.
1) Thiazolidinediones , like pioglitazone or rosiglitazone , which specifically target insulin resistance
2) Drugs, like albiglutide or dulaglutide , that mimick glucagon-like peptide-1 a hormone that promotes insulin secretion, and
3) Dipeptidyl peptidase 4 inhibitors, which increase GLP-1 levels, and lead to insulin secretion and lowering of blood sugar levels
Results
Learn More
Parkinson’s Gene May Impair How New Neurons Are Made Throughout Our Lifetime
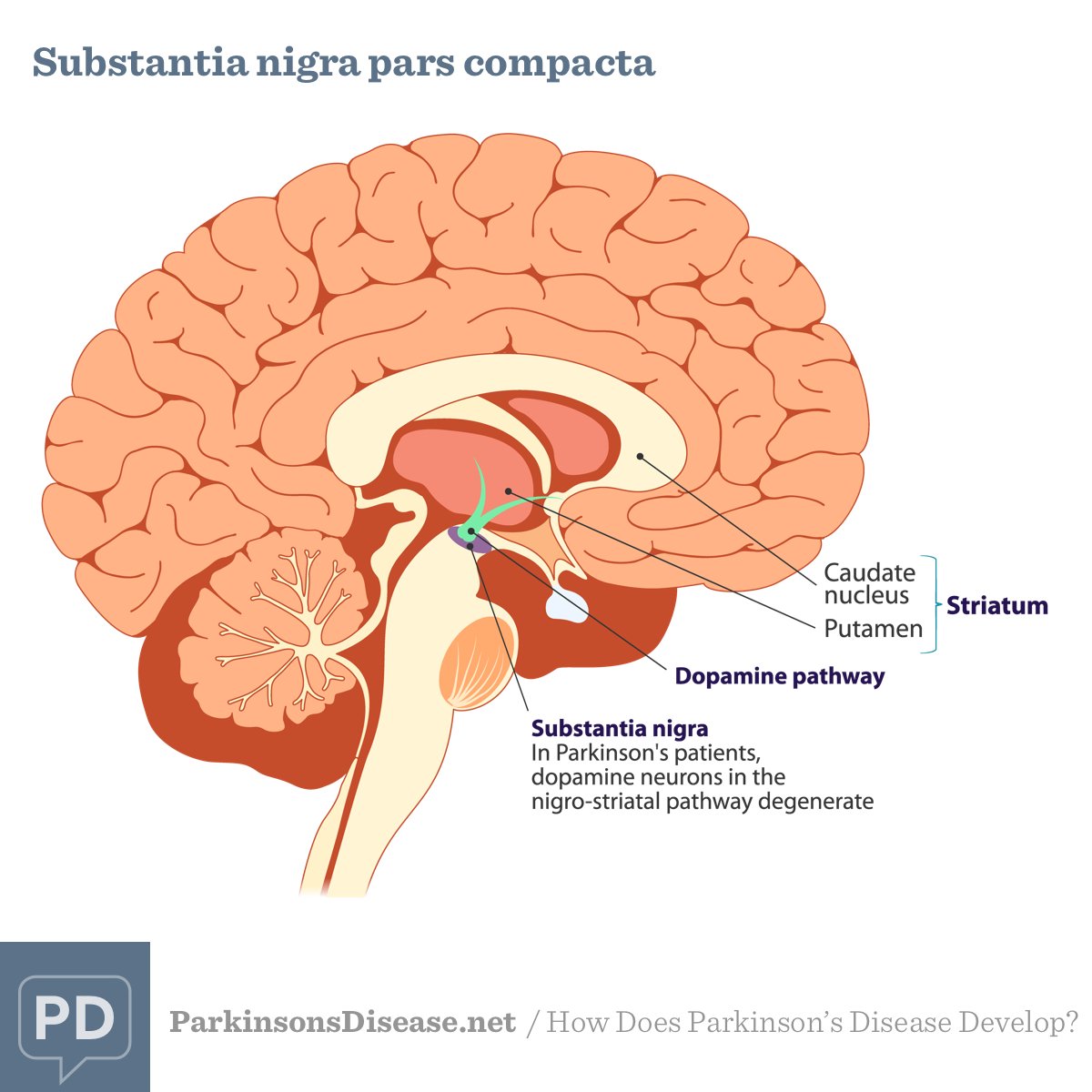
A gene defect linked to Parkinson’s disease may not only cause the early death of neurons, but also impair the process that generates neurons in the brain throughout our lifetime, a new study has revealed.
- A pioneering study, published in Scientific Reports, found that the Parkinson’s gene PINK1 is important for the generation of dopamine-producing neurons throughout life, and is not just responsible for the premature death of these neurons
- The international research, led by University of Sheffield’s Neuroscience Institute, used two model systems to examine how neurons are produced throughout our lifetime
- Parkinson’s disease is a relentlessly progressive neurodegenerative disorder that affects around 145,000 people in the UK
A gene defect linked to Parkinson’s disease may not only cause the early death of neurons, but also impair the process that generates neurons in the brain throughout our lifetime, a new study has revealed.
The international study, led by the University of Sheffield’s Neuroscience Institute, may have a significant impact on the future treatment of Parkinson’s patients who develop the illness due to PINK1 defect or similar gene defects.
The development of novel treatments and therapies to slow down disease progression, halt or reverse Parkinson’s may now focus on enhancing the generation of new dopamine-producing neurons, rather than just trying to protect these neurons from dying later.
Earlier Research On The Link Between Type 2 Diabetes And Parkinsons Disease
Some previous research has linked certain medications for type 2 diabetes to a lower risk of the development or progression of Parkinson’s disease.
A found Parkinson’s disease symptoms improved in participants who took exenatide, a diabetes drug in a family of medicines known as GLP1 agonists, and worsened when subjects took a placebo. Another , found that individuals with type 2 diabetes who took GLP1 agonists or another type of diabetes drugs known as DPP4 inhibitors had a lower risk of developing Parkinson’s disease.
Slightly elevated blood sugar or variations in blood sugar may contribute to the risk of Parkinson’s disease even in people without diabetes, according to a .
Age is the biggest risk factor for Parkinson’s disease, though, and genetics also account for up to 20 percent of the risk, Foltynie says.
RELATED: How to Keep Your Brain Healthy: A Conversation With Sanjay Gupta, MD
Links Between Insulin Resistance Metabolic Syndrome And Parkinsons Disease
Insulin resistance and metabolic syndrome are very common in the United States, but are also noted to have increased incidence in patients with Parkinson’s disease . Insulin resistance may precede the development of diabetes by many years, but with treatment, diabetes can be avoided.
Growing evidence now shows an association of insulin resistance and metabolic syndrome with worse symptoms and progression of Parkinson’s disease . Lima et al. have shown diabetes incidence in PD is associated with faster progression of both motor and cognitive symptoms.
The Association Between Type 2 Diabetes Mellitus And Parkinsons Disease
Article type: Review Article
Affiliations: Barts and The London School of Medicine, Queen Mary University of London, London, UK | Reta Lila Weston Institute of Neurological Studies, UCL Queen Square Institute of Neurology, London, UK | Department of Clinical and Movement Neurosciences, University College London Institute of Neurology, London, UK | Preventive Neurology Unit, Wolfson Institute of Preventive Medicine, Queen Mary University of London, London, UK
Correspondence: Correspondence to: Alastair J. Noyce, MRCP, PhD, Preventive Neurology Unit, Wolfson Institute of Preventive Medicine, Queen Mary University of London, Charterhouse Square, London EC1M 6BQ, UK. Tel.: +44 207 882 5841; E-mail: .
Keywords: Parkinson’s disease, type 2 diabetes mellitus, epidemiology, therapeutics, mechanisms
DOI: 10.3233/JPD-191900
Journal: Journal of Parkinson’s Disease, vol. 10, no. 3, pp. 775-789, 2020
Abstract
Insulin Dysregulation May Be Involved In Pathophysiology Of Pd And T2dm
Insulin receptors are expressed in the basal ganglia and in the substantia nigra , which are the areas of the brain most affected in patients with PD. Studies using rodent models have shown that insulin resistance may cause reduced expression of surface dopamine transporters in the striatum , reduced dopamine turnover , and reduced insulin-dependent dopamine release in the striatum . 1-methyl 4-phenyl 1,2,3,6-tetrahydropyridine is a toxin that induces parkinsonism by producing oxidative stress in dopaminergic neurons, resulting in mitochondrial dysfunction and cell death, and MPTP treated rodents are one of the most commonly used animal models for PD . MPTP-treated mice have been observed to have simultaneous increases in pancreatic and midbrain expression of pro-inflammatory cytokines and ?-synuclein, hinting at potential organ-specific links between PD and T2DM .
How Are We Using Stem Cells To Understand And Treat Parkinsons Disease
Stem cells are being used in multiple ways to understand the disease process and find new ways to potentially treat PD. By using stem cells in the lab to either model the disease by studying the affected neurons under the microscope or make healthy neurons to transplant into patients to replace the cells lost to the disease, stem cells are helping scientists discover new clues about how the disease is triggered and how it might one day be treated.
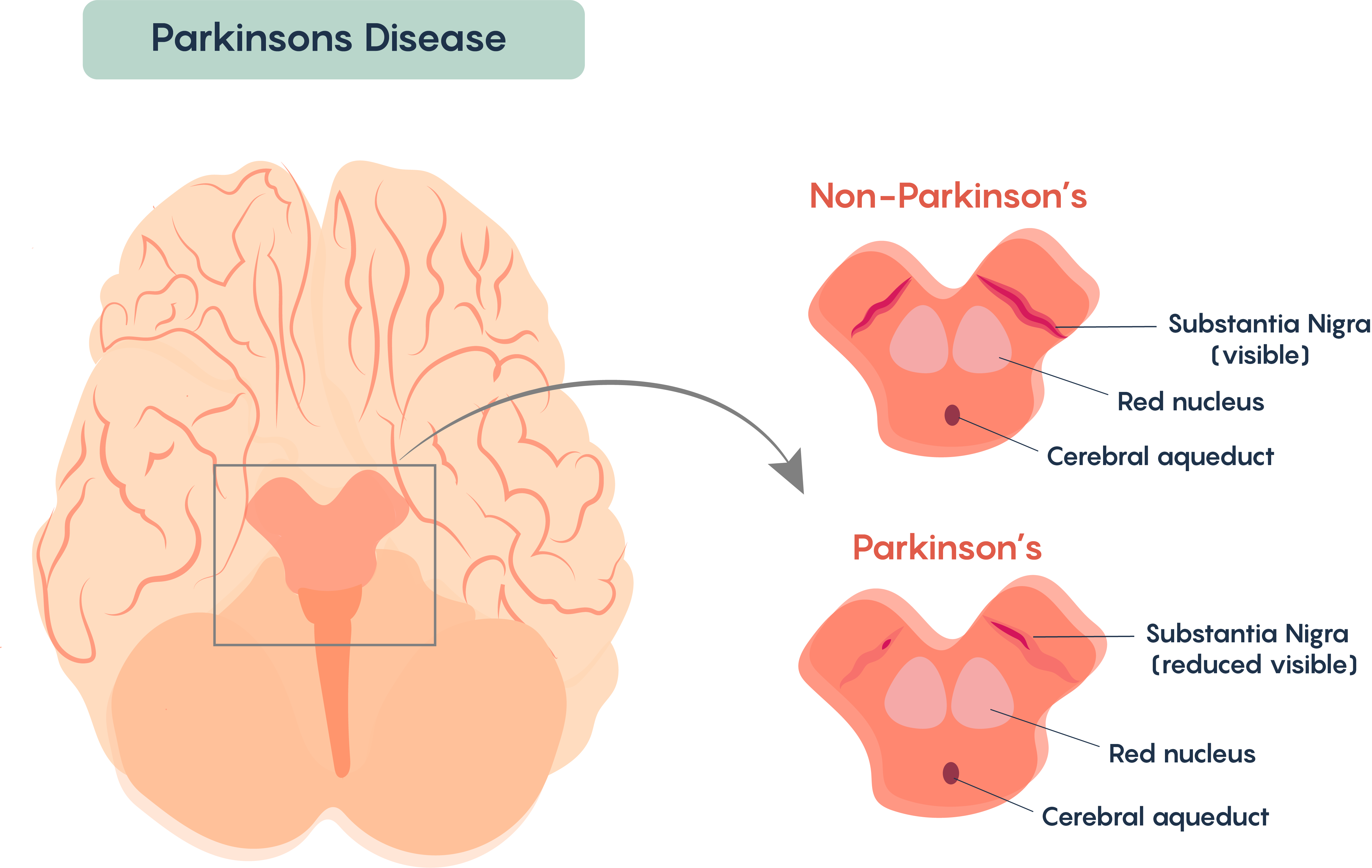
Parkinsons Disease And The Vulnerability Of The Nigrostriatal Pathway
PD is the prototype of movement disorders. In fact, the most prominent feature of PD is the presence of motor symptoms expressed in a majority of patients as a classical triad of resting tremor, bradykinesia, and rigidity . Non-dopaminergic motor features may also arise and result in falls, freezing of gait, speech impairment and difficulty in swallowing. Accompanying motor symptoms are equally disabling non-motor manifestations, such as psychiatric disturbances, dementia and autonomic failure .
The neuropathological hallmark of PD is the progressive and relentless degeneration of dopaminergic neurons located in the substantia nigra pars compacta. Idiopathic PD still composes approximately 90–95% of diagnoses that are not always faithfully represented by genetic forms in terms of symptomatology and pathophysiology .
Despite their apparent disparity, multiple mechanisms converge to cause the degeneration of nigrostriatal neurons and possibly contribute to oxidative stress to which these neurons may be more vulnerable . This concept dates back to the 1980s , but its popularity has waxed and waned at the rhythm of discoveries; nevertheless, the idea that metabolic oxidative insults may underlie the specific vulnerability of nigrostriatal neurons has lately gained momentum .
6. Last, they are endowed with a limited calcium-buffering capacity and scanty endogenous antioxidative defenses , consequent of their low expression of calbindin and glutathione , respectively.
Common Pathogenic Mechanisms Of Systemic And Brain Insulin Resistance
As epidemiological evidence for a link between PD and T2DM accumulates, parallel experimental evidence indicates potential overlap in disease mechanisms and pathways. Systemic insulin resistance has long been an established key feature of T2DM. Recently, studies have found that insulin resistance is present in the brain in neurodegenerative diseases such as Alzheimer’s disease and other dementias , and PD . Both systemic and local insulin resistance may drive pathology in the brain. Systemic insulin resistance may do so through hyperglycaemia and its consequences , microvascular disease, chronic inflammation, and dysfunction of the blood brain barrier, which may be compounded by associated comorbidities such as hypertension, dyslipidaemia and renal impairment . Local brain insulin resistance may act via protein deposition and aggregation, and failure of clearance mechanisms, independent of systemic insulin resistance .
Metformin Linked To Increased Risk Of Dementia And Parkinsons Disease
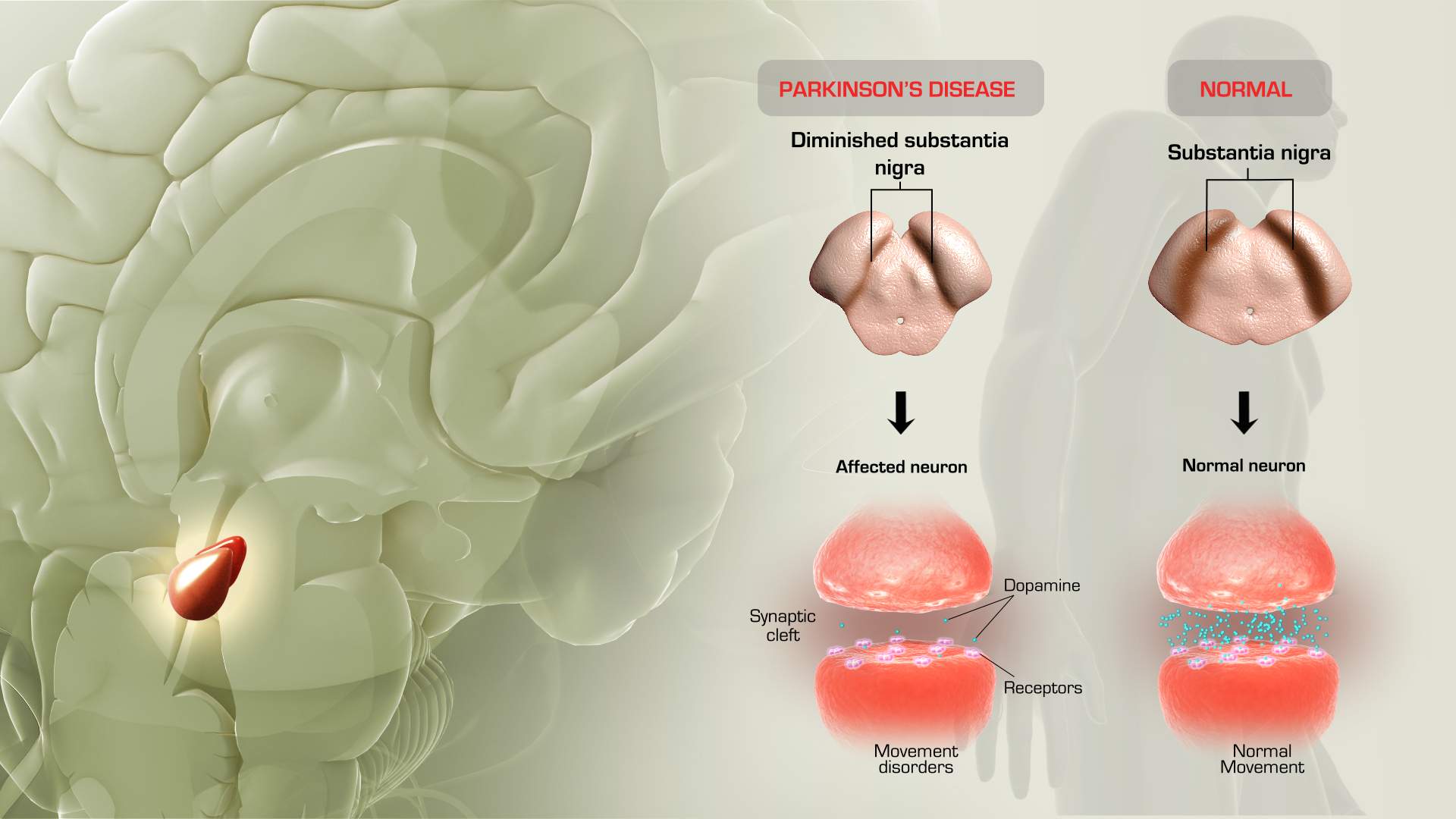
Study finds connection between duration of therapy in senior patients and development of neurodegenerative disease.
In 2011, the Journal of Alzheimer’s Disease published the findings of a large Taiwanese study showing a protective effect against development of dementia in diabetes patients who were given oral antidiabetic agents. The cohort of over 100,000 subjects included patients over 50 with type 2 diabetes, who were free of dementia at initiation, and received either or both metformin and a sulfonylurea. The results suggested that while T2D carries a two-fold increase in the risk of dementia, use of metformin, sulfonylureas, or both can reduce the risk by up to 35% over eight years. Medscape recently reported that at AD/PD 2017 , a group of Taiwanese neurologists presented the results of their own study looking at possible risk increases for Alzheimer’s and Parkinson’s in people with type 2 diabetes, citing uncertainty about the effects of metformin on the risk of developing neurodegenerative diseases.
Practice Pearls:
- Type 2 diabetes has long been understood to carry an increased risk of developing neurodegenerative diseases.
- Until recently, metformin and sulfonylurea use in T2D has been associated with a decrease in risk for Parkinson’s and Alzheimer’s diseases.
- A recent study suggests metformin use can increase the risk of Parkinson’s and Alzheimer’s, but there are also concerns over the validity of the findings due to study design flaws.
References:
How Type 2 Diabetes May Contribute To The Risk Of Parkinsons Disease
Although the analysis wasn’t designed to determine how type 2 diabetes might cause Parkinson’s disease to develop or progress, it’s possible that systemic inflammation present with type 2 diabetes may contribute to Parkinson’s disease, says Noyce.
Vascular disease that develops with type 2 diabetes may also lead to impaired blood flow to the brain that hastens the development of Parkinson’s disease, hypothesizes Emanuele Cereda, MD, PhD, of the clinical nutrition and dietetics unit at Fondazione IRCCS Policlinico San Matteo in Pavia, Italy, who was not involved in the current study.
Another possibility is that the same processes that cause diabetes also cause the nerve cell degeneration present in Parkinson’s disease, says Tom Foltynie, PhD, a professor of neurology at University College London in the United Kingdom.
In particular, insulin resistance, the body’s inability to respond normally to the hormone insulin, may be involved in both type 2 diabetes and Parkinson’s disease, says Dr. Foltynie, who also was not involved in the current research.
RELATED: Why Some Researchers Are Calling Alzheimer’s a ‘Type 3 Diabetes’
Pathophysiology Of Nigrostriatal Da Depletion In The Motor Circuit
These newer approaches have focused on dynamic features of neuronal activity changes in PD – such as oscillatory bursting and synchronization of discharge among neighboring neurons – rather than static features such as mean firing rates. Recordings in PD patients and primates with experimental parkinsonism reveal low-frequency oscillatory field potentials and rhythmic neuronal bursting in both STN and GPe.,,, Neurons in both structures show correlated discharge in the parkinsonian state. Effective symptomatic treatment with dopaminergic medication reduces or abolishes the low-frequency oscillatory activity as well as the correlations among neurons.
Epidemiological Evidence: Parkinsons Disease In Diabetic Patients
The first indications of a possible association between DM and PD date back to almost 60 years ago. Studies conducted in parallel showed that DM exacerbates the progression of motor and cognitive deficits in PD , and that non-diabetic parkinsonian patients present impaired glucose tolerance and hyperglycemia . Since then, these findings have been reiterated in parkinsonian patients medicated with dopamine-replacing therapy , but also in drug-naïve parkinsonian patients who still displayed higher-than-normal levels of fasting blood glucose, within the prediabetic diagnostic range . In addition, dietary habits featuring an elevated intake of high glycemic indexed carbohydrates have more recently been associated with greater odds of developing PD .
Despite the recent medical advances in teasing out the cause-effect relationship linking DM and PD, it is still unclear which of the multifarious metabolic changes that occur in DM trigger the nigrostriatal degeneration underlying PD. The two predominant lines of thought grant importance to the dimensions of defective insulin signaling and oxidative stress.
Rerouting Mechanisms: Polyol Pathway And Macromolecules Glycation
Aside from ROS generated through mitochondrial failure, hyperglycemia may trigger the onset of oxidative stress via pathways that dwell upstream the glycolytic enzyme GAPDH .
As previously discussed, free glucose is present in the cytosol of neurons and its concentrations rise during hyperglycemia leading to glucotoxicity. Under normal circumstances, hexokinase is mandated to transform available glucose into glucose 6-phosphate. However, due to substrate-mediated inhibition, in hyperglycemic conditions, it can only funnel a fraction of cytosolic glucose toward the glycolytic pathway before becoming saturated. Excess glucose is therefore consumed by other pathways and is constantly replenished by high extracellular levels. The principal compensatory outlets are the polyol pathway and macromolecule glycation which are two key pathways through which glucotoxicity manifests .
AGEs may also occur extracellularly or being directly secreted from the cells in which they are generated to induce oxidative stress and inflammation in a variety of cells . AGEs trigger oxidative stress by binding their cognate membrane receptor, the RAGE and activating downstream mediators know to induce oxidative stress, such as the inducible nitric oxide synthase . Nevertheless, despite AGEs being described for their ability to promote oxidative stress in peripheral tissues , their role in oxidative stress in PD remains to be elucidated.
Type 2 Diabetes Linked To Increased Risk Of Parkinsons Disease
Genetic data suggests there may be a direct relationship between type 2 diabetes and a higher risk of the movement disorder, as well as its progression, but more studies are needed.
Everyday Health
People managing type 2 diabetes and their doctors may need to have Parkinson’s disease on their radar, as new research sheds light on the possible connection between these two chronic health conditions.
For the review and meta-analysis, which was , researchers examined data combined from nine previous studies that followed individuals with type 2 diabetes over time to see if they developed Parkinson’s disease. They found type 2 diabetes associated with a 21 percent increased risk of Parkinson’s and with faster symptom progression. Parkinson’s causes muscle stiffness, tremors, impaired balance, and slow movement, in addition to cognitive and sleep issues, according to the National Institute on Aging.
Yet study authors could not account for the severity of participants’ type 2 diabetes, and they couldn’t determine the effect of diabetes drugs or quality of blood sugar management on Parkinson’s disease risk — two major limitations.
The research doesn’t prove that diabetes causes Parkinson’s, though researchers speculated the disease may contribute to this risk. They conducted a separate analysis of studies that identified common genetic variations present with the two conditions and found that type 2 diabetes directly increased the odds of developing Parkinson’s by 8 percent.
Bananas Also Contain Fiber Which May Reduce Blood Sugar Spikes
In addition to starch and sugar, a medium-sized banana contains 3 grams of fiber.
Everyone, including diabetics, should eat adequate amounts of dietary fiber due to its potential health benefits.
However, fiber is especially important for people with diabetes, as it can help slow the digestion and absorption of carbs .
This can reduce blood sugar spikes and improve overall blood sugar control .
One way of determining how a carb-containing food will affect blood sugars is by looking at its glycemic index .
The glycemic index ranks foods based on how much and how quickly they raise blood sugar levels.
The scores run from 0 to 100 with the following classifications:
Low GI: 55 or less.Medium GI: 56–69.High GI: 70–100.Diets based on low-GI foods are thought to be particularly good for people with type 2 diabetes .
This is because low-GI foods are absorbed more slowly and cause a more gradual rise in blood sugar levels, rather than large spikes.
Overall, bananas score between low and medium on the GI scale .
