Living With A Stimulator
Once the DBS has been programmed, you are sent home with instructions for adjusting your own stimulation. The handheld controller allows you turn the stimulator on and off, select programs, and adjust the strength of the stimulation. Most patients keep their DBS system turned on 24 hours day and night. Some patients with essential tremor can use it during the day and turn off the system before bedtime. Your doctor may alter the settings on follow-up visits if necessary.
If your DBS has a rechargeable battery, you will need to use a charging unit. On average charging time is 1 to 2 hours per week. You will have a choice of either a primary cell battery or a rechargeable unit and you should discuss this with you surgeon prior to surgery.
Just like a cardiac pacemaker, other devices such as cellular phones, pagers, microwaves, security doors, and anti theft sensors will not affect your stimulator. Be sure to carry your Implanted Device Identification card when flying, since the device is detected at airport security gates.
Dont Miss: Parkinsons And Immune System
Using A Different Preparation Of The Same Drug Treatment
Patients who usually receive levodopa can be given dispersible co-beneldopa via a nasogastric tube during lengthy surgery. A patient with severe Parkinsons disease undergoing liver resection was maintained without apparent exacerbation of his condition using this approach. Previous operations had resulted in marked rigidity and inability to swallow secretions in the same patient.15
This technique is unsuitable when evidence indicates paralytic ileus. Two cases of worsening parkinsonism have been reported in patients whose recovery was complicated by ileus despite receiving orally disintegrating co-careldopa.5
When switching from controlled release levodopa to dispersible co-beneldopa a dose reduction of around 30% is suggested due to lower bioavailability in controlled release form.1617
What Are The Results
Successful DBS is related to 1) appropriate patient selection, 2) appropriate selection of the brain area for stimulation, 3) precise positioning of the electrode during surgery, and 4) experienced programming and medication management.
For Parkinson’s disease, DBS of the subthalamic nucleus improves the symptoms of slowness, tremor, and rigidity in about 70% of patients . Most people are able to reduce their medications and lessen their side effects, including dyskinesias. It has also been shown to be superior in long term management of symptoms than medications .
For essential tremor, DBS of the thalamus may significantly reduce hand tremor in 60 to 90% of patients and may improve head and voice tremor.
DBS of the globus pallidus is most useful in treatment of dyskinesias , dystonias, as well as other tremors. For dystonia, DBS of the GPi may be the only effective treatment for debilitating symptoms. Though recent studies show little difference between GPi-DBS and STN-DBS.
Patients report other benefits of DBS. For example, better sleep, more involvement in physical activity, and improved quality of life .
Research suggests that DBS may “protect” or slow the Parkinson’s disease process .
Don’t Miss: Parkinson’s Tremor At Rest
Managing Parkinsons Disease During Surgery
- Accepted 16 September 2010
People with Parkinsons disease undergoing surgery are at increased risk owing to their condition and to potential omission of medication. This article looks at what doctors need to consider in this situation and how specialist assistance can be accessed
Parkinsons disease is a common condition affecting over 100000 people in the UK.1 People with Parkinsons disease who undergo surgery have increased mortality and longer hospital stays than people without this condition.2 Missing dopaminergic medication during a period of perioperative starvation can result in life threatening complications.34 This problem can be further compounded if the absorption of drugs is impaired.5 Recognising these hazards is the first step in an approach that may reduce the risk of suboptimal medication faced by people with Parkinsons disease, using three principles.
-
Advance planning
-
Advice from specialists.
Unplanned Surgery And Complicated Elective Surgery

Specialist preoperative advice cannot always be obtained, particularly for urgent surgery. To allow a treatment plan to be enacted at any time, we recommend that hospitals develop a protocol for this situation. We propose a flowchart to guide a non-specialist until more specific advice can be obtained from the movement disorder team.89 Clinicians who are unfamiliar with medication for Parkinsons disease may require assistance from a pharmacist.
Algorithm for estimating parenteral doses of drugs for Parkinsons disease.
Recommended Reading: Parkinson’s And Seborrheic Dermatitis
What It Feels Like: Deep Brain Stimulation For Parkinsons Disease
Emma Jones,
Manzil Bacchus was diagnosed with Parkinsons disease in 2008. Initially, he was told that there was nothing he could do to stop the progression of the progressive nervous system disorder that affects movement. Manzil and his wife, Sadia, continued to manage the increasingly worrisome symptoms which included tremors and the growing amount of medication that he had to take each day.
After Manzil was diagnosed with colitis, a chronic digestive disease that has been linked to Parkinsons, the family began investigating a new surgical option. In deep brain stimulation , electrodes are inserted in regions of the brain that have been affected by the Parkinsons, with an impulse generator battery also inserted in another area of the body. When turned on, the electrodes send gentle electrical pulses to help improve symptoms like tremors and motor control.
Manzil and Sadia sat down with Healthing to talk about Manzils experience having brain surgery during the COVID-19 pandemic and the difference that deep brain stimulation has made to his quality of life.
When was Manzil diagnosed with Parkinsons?Manzil: Back in 2007, my hands started shaking. I was volunteering at a mosque, helping people to take a pilgrimage in Saudi Arabia, helping with their application for visas and stuff like that. One of the girls noticed my hand shaking and asked why. I thought I was just tired. After that, I also noticed that my left foot was dragging when I walked.
Sadia: Sadia: Sadia:
Neurobehavioral And Psychiatric Disorders
Parkinsons disease is a slowly progressing, degenerative disease that is the most common form of Parkinsonism, a group of motor system disorders.
For some time it was incorrectly believed that Parkinsons disease disappeared after the introduction of levodopa in the 1960s. In fact, about 50,000 Americans are diagnosed with Parkinson’s disease each year, with more than half a million Americans affected at any one time. Further, more people suffer from Parkinsons disease than multiple sclerosis, muscular dystrophy, and amyotrophic lateral sclerosis combined.
Parkinsons disease is chronic , and progressive .
Although the disease may appear in younger patients , it usually affects people in late middle age. The disease affects men and women in almost equal numbers. It is not contagious, nor is it likely passed on from generation to generation.
Don’t Miss: Can Parkinson Tremors Be Controlled
Potential Complications Of Missing Medication
The consequences of missing Parkinsons medication can vary enormously. Some people can tolerate a missed tablet without experiencing any major effects. Others become immobile. However, in some situations, missing dopaminergic medication can precipitate a condition known as neuroleptic malignant like syndrome, associated with fever, confusion, raised concentrations of muscle enzyme, and even death. This syndrome is most common in people with more severe Parkinsons symptoms and those on the largest doses of levodopa.6
Surgery To Implant The Deep Brain Stimulation Device
Deep brain stimulation requires the surgical implantation of an electrical device into the brain. A neurosurgeon uses imaging scans to pinpoint the right spot in the brain for implanting the electrode.
When surgeons have determined the correct location, they create a small opening in the skull and insert a thin, insulated wire, through which they insert the electrode. Surgery to implant the electrode takes about four hours and requires general anesthesia. You may then stay overnight in the hospital for observation.
The next day, doctors perform the second part of the surgery, which involves connecting the insulated wire to a battery-operated pulse generator that is implanted under the skin near the collarbone. Most people can return home after this procedure.
Several days after the surgery, you meet with your neurologist, who programs the pulse generator. Pushing a button on an external remote control sends electrical impulses from the pulse generator to the electrode in the brain.
People who use deep brain stimulation work closely with their neurologist to find the combination of settings that best controls their symptoms. After several visits, they are able to control the strength of the electrical impulses on their own. Following this adjustment period, most people require only occasional maintenance visits.
You May Like: Can A Person Have Parkinson’s Without Tremors
How Deep Brain Stimulation Works
Exactly how DBS works is not completely understood, but many experts believe it regulates abnormal electrical signaling patterns in the brain. To control normal movement and other functions, brain cells communicate with each other using electrical signals. In Parkinson’s disease, these signals become irregular and uncoordinated, which leads to motor symptoms. DBS may interrupt the irregular signaling patterns so cells can communicate more smoothly and symptoms lessen.
Ological Limitations Of Surgery Trials
The included trials all had methodological limitations common to non-analytical study designs. Firstly, none of the included trials were randomised into surgical or non-surgical intervention groups. Secondly, none of the trials were performed under blinded conditions, either single or double. None of the trials were controlled with a of non-surgical patients for longitudinal comparison over time.
There was also a general lack of inclusion/exclusion criteria, which could lead to pre-selected patient populations, lack of multi-centre comparative results analysis, and lack of calculations. The mean of most trials was 712 months and the patient population tended to be younger with an average age of approximately 60 years.
What is the effectiveness and safety of any procedure versus standard medical therapy in the treatment of motor complications in patients with ?
Recommended Reading: How Does Parkinson’s Disease Develop
Resources For More Information
- Surgical option a potential life-changer for patients with OCD: Read and watch Erins story as she, a lively 21-year-old woman, fought her battle with OCD. This article explores how deep brain stimulation gave Erin her life back. The procedure was the first of its kind performed at Albany Medical Center the only facility offering this treatment between New York and Boston. In Erins own words, Now, I can be who I really am and tell people my story and hopefully inspire people and help people along the way.
- Karen and Jims Story: A Shared Journey of Life, Love and DBS: Read about Karen and Jim. They were each diagnosed with Parkinsons before they met. Follow them on their journey as they fall in love after meeting each other from an online support group. See how they embraced each other and DBS.
- Kays Story A Parkinsons Disease Patient: Read about Kay, a 68-year-old woman suffering from Parkinsons disease. The article and video explore how DBS helped her regain her life. In Kays own words, Its like I had been turned on again. It was like a miracle.
Dont Miss: How To Tell Difference Between Parkinsons And Essential Tremor
When To Consider Surgical Therapies In Parkinsons Disease
Oral medications, including dopaminergic and nondopaminergic options, are the mainstay of management in Parkinsons disease . Early in the disease course, medical management is effective in controlling motor symptoms and improving quality of life in a majority of patients. However, with disease progression and chronic use of dopaminergic therapies, patients can develop motor fluctuations and dyskinesia. For some patients, even early in the disease course, medication side effects can limit their therapeutic effectiveness, or tremor may be medication refractory. Surgical and other advanced treatment options should therefore be considered for patients whose symptoms cannot be adequately managed by oral medications alone. Prior to considering such advanced therapies, generally, the dose and frequency of dopaminergic medications should be optimized.
The currently available advanced treatments include deep brain stimulation , ablative or lesioning procedures, and dopaminergic medication infusion devices . These therapies have one of the following two effects: improvement of motor symptoms through targeted stimulation/ablation of the motor circuit and continuous dopaminergic medication delivery. The decision to pursue advanced therapies is typically guided by unsatisfactory control of motor symptoms, although both positive and negative effects on non-motor symptoms may often be observed.
Fig. 1
Read Also: What Are First Symptoms Of Parkinson’s
Who Is A Candidate
You may be a candidate for DBS if you have:
- a movement disorder with worsening symptoms and your medications have begun to lose effectiveness.
- troubling “off” periods when your medication wears off before the next dose can be taken.
- troubling “on” periods when you develop medication-induced dyskinesias .
DBS may not be an option if you have severe untreated depression, advanced dementia, or if you have symptoms that are not typical for Parkinson’s.
DBS can help treat symptoms caused by:
- Parkinson’s disease: tremor, rigidity, and slowness of movement caused by the death of dopamine-producing nerve cells responsible for relaying messages that control body movement.
- Essential tremor: involuntary rhythmic tremors of the hands and arms, occurring both at rest and during purposeful movement. Also may affect the head in a “no-no” motion.
- Dystonia: involuntary movements and prolonged muscle contraction, resulting in twisting or writhing body motions, tremor, and abnormal posture. May involve the entire body, or only an isolated area. Spasms can often be suppressed by “sensory tricks,” such as touching the face, eyebrows, or hands.
After your evaluation and videotaping is complete, your case will be discussed at a conference with multiple physicians, nurses, and surgeons. The team discusses the best treatment plan for each patient. If the team agrees that you are a good candidate for DBS, you will be contacted to schedule an appointment with a neurosurgeon.
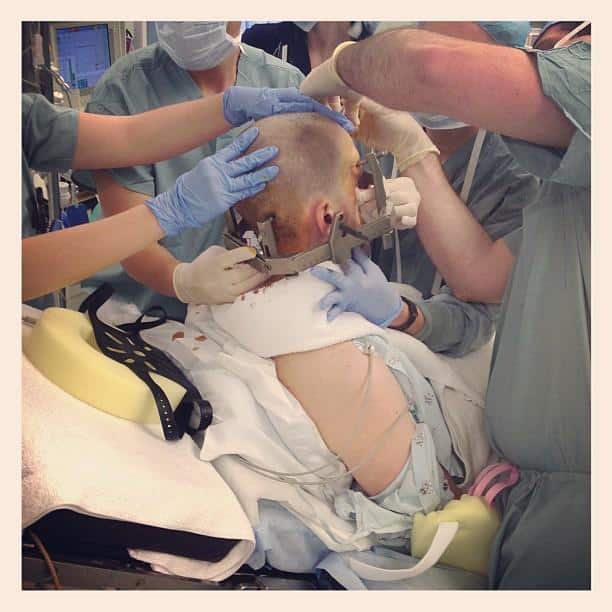
Will I Be Asleep During The Entire Procedure
You will be sedated or receive local anesthesia for part of the procedure, may or may not be awake for lead and electrode placement, and will be asleep when the impulse generator is implanted. To provide more details:
- A local anesthetic is applied to areas of the head where pins or screws are used to secure the head frame and sedation is given.
- You will be sedated during the beginning of the procedure, while the surgical team is opening the skin and drilling the opening in the skull for placement of the lead.
- Most patients will be awake for lead and electrode placement. This part of the procedure is not painful, as the brain does not feel pain. Being awake allows the surgical team to interact with you when testing the effects of the stimulation. However, some patients who cannot tolerate the procedure while awake can have the electrode and lead placed under general anesthesia. The lead placement is guided in real time by magnetic resonance imaging. The procedure is performed in a special MR-equipped operation room.
- Implantation of the pulse generator in the chest and connection of the leads from the device to the lead in the brain is usually done under general anesthesia.
Read Also: Is Beer Good For Parkinson’s
Research To Improve Deep Brain Stimulation
Researchers are working to improve upon existing DBS devices and methods to help treat more symptoms and more people. Some researchers are putting electrodes in a different area of the brain the pedunculopontine nucleus to treat walking and balance problems that don’t typically improve with present-day DBS. Others are developing a “smart” DBS device that can record a person’s unique brain signals and deliver electrical stimulation only when needed, such as when symptoms return, rather than continuously, as the current systems do. This could help reduce side effects such as numbness and weakness and lengthen the battery life of the neurostimulator, which would result in a longer time between battery replacement procedures.
Scientists also are planning to test deep brain stimulation in the first years after a Parkinson’s diagnosis to see if the therapy may slow or stop disease progression. Testing in Parkinson’s models showed the therapy may help protect brain cells, and a small human trial showed motor symptoms improved after early-stage DBS.
What Particular Risks Are Faced
Some of the risks relate to Parkinsons disease itself and others to the effects of omitting medication. A retrospective cohort study of 234 people with Parkinsons disease and 40979 controls undergoing major abdominal surgery found a higher incidence of aspiration pneumonia, bacterial infection, and urinary tract infection in the group with Parkinsons.2
Case reports have described other perioperative complications in people with Parkinsons disease, including postoperative respiratory failure and postextubation laryngospasm. Other reports mention exacerbation of Parkinsons symptoms during surgery and a case of severe neuroleptic malignant like syndrome precipitated by perioperative starvation for coronary artery bypass grafting.34
Access to the correct medication at the right time remains a problem for people with Parkinsons disease when they are admitted to hospital. This concern has led to a national awareness campaign from the charity Parkinsons UK.7 Lack of awareness of the importance of maintaining medication at the time of surgery places patients at increased risk.
You May Like: New Treatments For Parkinson’s Disease 2021
Parkinsons Disease Brain Surgery: What To Expect
Parkinsons disease brain surgery can either be done under general anesthetic or with you awake and alert with a local anesthetic to numb your skull. Either way, you shouldn’t feel anything during the procedure because there are no pain receptors in the brain. The surgery is performed in two parts: one for the insertion of electrodes into your brain, and one for the placement of the neurotransmitter in your chest.
After surgery, your device will be turned on at the doctors office and you will be given a remote control to take home with you. From then onwards, your doctor will work with you to help you manage your symptoms. You may also decide to turn the device off at certain times. It could take a few months to find the right setting for you.
