Faqs: Genetics & Parkinsons
If I have Parkinson’s disease will my child get it too? Will I inherit Parkinson’s if my parent or grandparent has it?
Most people with Parkinson’s have no known genetic link. Their children will likely never develop Parkinson’s. There are some known genetic variations that increase the risk of getting Parkinson’s, but most people with these variations do not get Parkinson’s. Like many other diseases, Parkinson’s is a result of a complex interaction between genes and environmental factors.
In a small number of people , Parkinson’s is inherited and can affect multiple family members. Their children may have a higher risk of developing Parkinson’s. However, there is no guarantee they will develop PD.
What if my genetic test is positive for a Parkinson’s gene?
Scientists have identified several genetic mutations that can increase the risk of developing Parkinson’s. If someone tests positive for a mutation in a Parkinson’s gene, it does not necessarily mean they will develop PD. Some people who have mutations in the genes associated with Parkinson’s never develop PD. A person may inherit a hereditary genetic mutation that increases their risk for Parkinson’s however, they may also inherit other genes, be exposed to environmental factors or have lifestyle choices that offset the risk. Genetic testing is currently available for the following genes related to Parkinson’s: GBA, PARK7, SNCA, LRRK2, parkin and PINK1.
What can I do with my genetic test results?
Advancing Age And Parkinsons Disease
Age is perhaps the biggest risk factor for the onset of Parkinsons disease. The average age at which people will develop this movement disorder is 60. This is not usually something that affects younger people. The brain ages as people get older.
Even without external factors, cells in the substantia nigra can die on their own as an individual ages, causing symptoms to develop as the person gets older.
Genes Connected To Parkinson’s
SNCA
In 1997, researchers at the National Institutes of Health made the first Parkinsons genetic connection, discovering that mutations in the SNCA gene were common in several families who had many members with Parkinsons. While mutations in this gene are rare, they have taught us invaluable information about PD.
The SNCA gene instructs the body to make the protein alpha-synuclein, which for reasons that are not yet known, clumps in the brain cells of people with PD. Study of the SNCA gene led to this understanding that applies to nearly all people with the disease. Since then, alpha-synuclein has been a major target of new drugs for Parkinsons.
LRRK2
Another gene that plays a role in Parkinsons is LRRK2, which accounts for one to two percent of all cases. For people of particular ethnic backgrounds Ashkenazi Jewish and North African Berber descent mutations in the LRRK2 gene account for a much greater number of cases than in the general PD population. Mutations in LRRK2 lead to too much activity of the LRRK2 protein, though recent research indicates people without a mutation can also have overactivity of LRRK2.
Shake It Up are funding several studies into LRRK2 and you can learn more about these here.GBA
PRKN
You May Like: How Is Parkinson’s Disease Inherited
Protein And Membrane Trafficking
SNPs within loci associated with protein and membrane trafficking recognized as PD susceptibility factors by GWASs are listed in Table 3. The genes within this group are involved in processes, such as clathrin-mediated vesicular transport, trafficking, and fusing of synaptic vesicles, clearance of Golgi-derived vesicles, endo- and exocytosis and protein sorting.
Table 3. SNPs within loci associated with protein and membrane trafficking recognized as PD susceptibility factors by GWASs.
TMEM175/GAK/DGKQ
A haplotype block TMEM175/GAK/DGKQ was first associated with sporadic PD in the GWAS performed by Hamza et al. after it was already confirmed to be associated with the familial form of the disease . This locus was reevaluated several times by different research groups and also showed positive results in other GWASs and meta-analyses of GWASs. The most replicated SNP is an intron variant rs11248060, others were found in individual studies only .
Less is known about DGKQ however, it is expressed in the brain where it may be involved in the phosphatidylinositol and lipid signaling . DGK proteins are also known to affect Ca2+ signaling as well as the trafficking and fusing of synaptic vesicles at nerve terminals in the central nervous system .
NUCKS1/RAB7L1
LRRK2
INPP5F
BCKDK/STX1B
VPS13C
What Should You Know About At
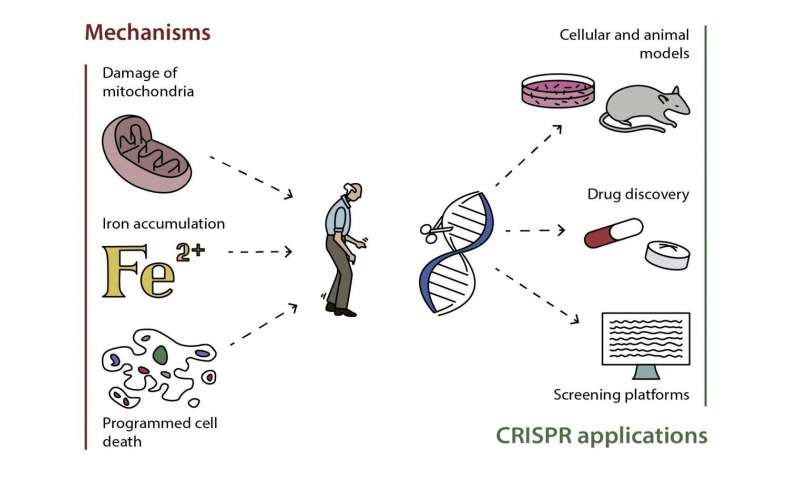
Genetic tests are not a substitute for a Parkinson’s diagnosis. Most at-home genetic tests do not provide genetic counseling services to help interpret the results. Always consult with a genetic counselor and your doctor before and after taking a genetic test. Most at-home genetic tests check for a limited number of gene mutations associated with Parkinson’s. This can be misleading since these tests may not be comprehensive.
Since scientists are still discovering more PD-associated genes, it is important to consult your doctor about comprehensive genetic testing options, like the PD GENEration study, which provides a free comprehensive genetic test identifying all possible variants in the two most common PD genes.
Even if you or a loved-one test positive for a Parkinson’s gene, it does not mean either of you will develop it. Having a genetic mutation only means that you may be at increased risk to develop PD. Environmental factors and lifestyle choices will help determine whether someone will get Parkinson’s.
You May Like: Sporadic Parkinson’s Disease
Susceptibility Genes For Pd
Although the study of large families segregating Mendelian forms of PD have provided substantial insight regarding the etiology and pathogenesis of PD, mutations in these genes have been found in fewer than 5% of all PD patients. Previously published data suggest that the risk of PD among the first-degree relatives of an affected individual is 2 to 14 times higher than the risk in the general population. These data would suggest that additional loci contribute to the risk of PD.
Several different approaches have been used to identify these additional loci. Several research groups have identified multiplex PD families, typically those with at least a sibling pair with disease, and have performed a whole genome linkage screening to identify chromosomal regions linked to the risk of PD or the age of PD onset. Chromosomes 5 and X have appeared in multiple linkage studies. Analyses combining data from two studies was not able to replicate linkage to Chromosome 5. Additional analyses on the X chromosome have identified several candidate genes however, none have been verified.
The evaluation of particular candidate genes has led to the identification of several susceptibility genes however, most have failed to consistently replicate in other populations. An exhaustive review of all genes analyzed as a candidate gene for PD is beyond the scope of this review. Rather, we have summarized the genes that have been the subject of the most intense focus in recent years.
NR4A2
SNCAIP
Is Parkinson’s Considered A Hereditary Disease
Nope, Parkinson’s isn’t considered a hereditary disease in most people. According to the Parkinson’s Foundation, about 90% of people with the disease have no genetic link.
And while there are some genetic markers for Parkinson’s, they don’t guarantee that a person will get the disease. A genetic mutation is just one of several risk factors for Parkinson’s disease. There may also be lifestyle choices and environmental factors involved in the development of the disease. In fact, most people with Parkinson’s disease aren’t aware of any other family member with the condition, according to the National Human Genome Research Institute .
RELATED: Coffee May Protect Against Dementia and Parkinson’s
In rare cases, Parkinson’s disease can run in families. When three or more relatives get the diseaseespecially under the age of 50, which is considered early onsetexperts believe members of the family may have a genetic predisposition to Parkinson’s.
You May Like: What Are The Four Cardinal Signs Of Parkinson’s Disease
What Raises Someone’s Risk For Parkinson’s
It’s a complex picture, but you may be more likely to get Parkinson’s based on:
Age. Since it mostly affects people 60 and older, your risk goes up as the years go by.
Family history. If your parent, brother, or sister has it, you’re a little more likely to get it.
Job. Some types of work, like farming or factory jobs, can cause you to have contact with chemicals linked to Parkinson’s.
Race. It shows up more often in white people than other groups.
Serious head injury. If you hit your head hard enough to lose consciousness or forget things as a result of it, you may be more likely to get Parkinson’s later in life.
Gender. Men get it more than women. Doctors aren’t sure why.
Where you live. People in rural areas seem to get it more often, which may be tied to chemicals used in farming.
What Causes Parkinsons Disease
Developing Parkinsons disease is not something that often happens on its own. Of course, there are genetic factors that could play a part in this neurological disorder. However, it is a combination of genetic and environmental factors that could lead to this disease.
In some cases, we have seen recent lawsuits that allege that exposure to certain chemicals has caused Parkinsons disease. Here, we will focus on how people could develop Parkinsons. That could give you an idea of whether you may have a possible lawsuit.
You May Like: Does Parkinson’s Cause Memory Issues
Genes Linked To Parkinsons Disease
Theres a long list of genes known to contribute to Parkinsons, and there may be many more yet to be discovered. Here are some of the main players:
SNCA: SNCA makes the protein alpha-synuclein. In brain cells of individuals with Parkinsons disease, this protein gathers in clumps called Lewy bodies. Mutations in the SNCA gene occur in early-onset Parkinsons disease.
PARK2: The PARK2 gene makes the protein parkin, which normally helps cells break down and recycle proteins.
PARK7: Mutations in this gene cause a rare form of early-onset Parkinsons disease. The PARK7 gene makes the protein DJ-1, which protects against mitochondrial stress.
PINK1: The protein made by PINK1 is a protein kinase that protects mitochondria from stress. PINK1 mutations occur in early-onset Parkinsons disease.
LRRK2: The protein made by LRRK2 is also a protein kinase. Mutations in the LRRK2 gene have been linked to late-onset Parkinsons disease.
Among inherited cases of Parkinsons, the inheritance patterns differ depending on the genes involved. If the LRRK2 or SNCA genes are involved, Parkinsons is likely inherited from just one parent. Thats called an autosomal dominant pattern, which is when you only need one copy of a gene to be altered for the disorder to happen.
What Else Do We Know
As scientists try to learn what’s at the root of Parkinson’s, they’re looking far and wide to pick up clues where they can.
They’ve found that people with Parkinson’s tend to have something called Lewy bodies in their brain. These are unusual clumps of a protein called alpha-synuclein. The protein itself is normal, but the clumps are not. And they’re found in parts of the brain that affect sleep and sense of smell, which could explain some symptoms of Parkinson’s not related to movement.
Your gut may also have a part in it, as some of its cells make dopamine, too. Some doctors think that this might be where the earliest signs of Parkinson’s show up, but that idea needs more research.
You May Like: What Are The Four Cardinal Signs Of Parkinson’s Disease
Linking Familial And Sporadic Forms Of The Disease
Besides identifying several risk loci for sporadic PD, large population-based GWAS and meta-analyses have also established that disease linked common variability exists at loci known to contain rare causal mutations. These include SNCA, LRRK2, GBA and VPS13C, previously associated with Mendelian forms of PD thus, suggesting a link between familial and sporadic forms of disease. These loci harboring both rare large effect and common smaller effect variants are known as pleomorphic risk loci . Therefore, both the CDCV hypothesis and the multiple rare variant hypothesis are not mutually exclusive. Instead, various disease-related genetic mechanisms may coexist at the same locus, each influencing disease through different biological effects on a single gene. For example, SNCA locus has been linked to PD etiology through: duplications and triplications with a clear gene-dose effect coding mutations causing early-onset PD familial cases different association signals in GWAS illustrating the presence of low-effect common variability in this locus and non-coding risk/protective variants .
The Phss And Model Performance
The Cox stepwise regression framework identified 71 SNPs that met criteria for inclusion in the final PHS model . A graphical comparison between stratified KaplanâMeier estimations and Cox proportional hazard models indicated that the proportional hazard assumption holds for the final model . Notably, several SNPs were included from loci that have also been reported to be individually associated with age at onset, including SNCA and TMEM175 . PHS was normally distributed and showed a strong association with age-related PD incidence in the test data . Stratified KaplanâMeier estimates are shown in Figure , demonstrating the effect of PHSs on age at onset in the test data. The hazard ratio comparing the highest to the lowest risk deciles was 3.78 after sample weight correction.
FIG 2
Also Check: Mucuna Pruriens Parkinson
Parkinson’s Disease Genetic Influence
Recent developments in research gene research has found that genetic influence plays a large role in Parkinson’s disease. Five main genes that are believed to contribute to the disease have been identified and located. These include alpha-synuclein, Parkin, Ubiquitin carboxyl-terminal hydrolase, DJ-1 and SCA2. It has been found that mutations of these genes are some of the underlying causes. In some causes there can be multiple mutations in one gene. The effects of some of these mutations are now understood.
Researchers suspect that genes associated with the late onset of Parkinson’s Disease are susceptibility genes rather than causal genes. It is thought that environmental factors act on these gene, consequently leading to Parkinson’s disease. But the mechanism in which they do so is not yet known. Researchers believe that if they can work out this mechanism, they can disrupt it in some way, and therefore halt the onset of the disease.
The general consensus among researchers is that both genetic influence and environmental factors lead to the onset of Parkinson’s Disease. The mechanisms of the genetic influence of Parkinson’s Disease are still to be understood and much more research is required.
The content of this web site is intended to convey general educational information and should not be relied upon as a substitute for professional healthcare advice. More information.
Other Factors Influencing Parkinson’s Disease Risk
Other factors besides genetics can influence someone’s chances of developing Parkinson’s disease, including:
- Age: The risk of developing Parkinson’s disease increases as a person ages.
- Sex: Males have a higher chance of developing Parkinson’s disease than females.
- Family history: First-degree relatives of an individual with Parkinson’s disease have a higher chance of developing Parkinson’s disease.
- Exposure to certain chemicals increases the risk of developing Parkinson’s disease.
Read Also: Can Parkinson’s Run In The Family
Can Parkinsons Be Passed From Parent To Child
Its rare for Parkinsons disease to be passed down from parent to child. Most cases of Parkinsons arent hereditary. But people who get early-onset Parkinsons disease are more likely to have inherited it.
Having a family history of Parkinsons disease may increase the risk that youll get it. This means that having a parent or sibling with Parkinsons slightly increases the risk.
In most cases, the cause of Parkinsons disease remains unknown. But researchers have identified multiple risk factors that can increase your chances of getting this disease.
Risk factors for Parkinsons disease include:
- mutations in specific genes associated with Parkinsons
- having a family history of Parkinsons or a first-degree family member with Parkinsons
- being older, especially above the age of 60
- exposure to herbicides and pesticides
- being assigned male at birth
- history of brain injury
Are You At Higher Risk
It is relatively rare for a parent to pass down Parkinsons to a child. However, people who get early-onset Parkinsons are more likely to have inherited it.
In most cases, the cause of Parkinsons is unknown, although the most common risk factors include:
- Age
- Male gender
- Mutations in specific genes associated with Parkinsons
- A family history of Parkinsons
- Exposure to herbicides or pesticides
The only way to truly determine if you or a member of your family is carrying one of the genes associated with the disease is to participate in genetic testing. These tests can identify if you have an associated gene marker and how likely it is that it will be passed down to your children.
While there isnt a way to fully prevent Parkinsons disease, you can certainly be proactive, Dr. Hanna says. Genetic testing and counseling will allow you to discover if you do have a mutated gene, how likely it is that it will be passed down to your children, as well as the best treatment options for your diagnosis.
You May Like: What Is The Life Expectancy Of Someone With Parkinson’s Disease
Inferring Causal Relationships Across Phenotypic Traits And Exploring Shared Polygenic Risk
A corollary of abundant GWA data is the ability to test other traits that can predispose or protect individuals to disease. Access to large amounts of GWA data affords the opportunity to explore causal inferences between myriad traits and PD through Mendelian Randomization , the gold standard approach for causality in genetic studies that sits at the interface between observational epidemiology and interventional trials . MR has emerged as a statistical approach that uses genetic data in the form of SNPs to study whether an exposure exerts a causal effect in an outcome. This addresses the question of whether an observational association between a risk or protective factor and a disease of interest is consistent with a causal effect by focusing usually only on genome-wide significant SNPs. Put simply, genetic variants that explain variation in a certain exposure can be used as proxies to determine how a change in that exposure might influence a disease outcome. One of the key strengths of this method is that it relies on genetic variants that remain constant over the lifespan of an individual, are randomized during gametogenesis and fixed at conception, which means that genetic variants are not associated with all the confounder factors that affect an observational study.
In the PD field, two-sample MR has been applied considering both targeted exploration of causal associations, as well as hypothesis-generating approaches in a large-scale, high-throughput, unbiased manner.
