Tms May Ease Bradykinesia Rigidity In Parkinson’s
Megan Brooks
New research shows that high-frequency repetitive transcranial magnetic stimulation over bilateral motor cortex improves symptoms of bradykinesia and rigidity in patients with Parkinson’s disease .
This finding highlights the potential for developing targeted rehabilitation for specific motor symptoms based on specific cortical regions, the investigators say.
“Stimulation of the motor area can help Parkinson’s symptoms, and this can be sustained over time. Knowing which symptoms are helped by TMS may be important to define better the target area,” study investigator Alessandro Di Rocco, MD, executive director, The Fresco Institute for Parkinson’s & Movement Disorders at NYU Langone Medical Center in New York City, told Medscape Medical News.
The findings, from a secondary subset analysis of the Magnetic Stimulation for the Treatment of Motor and Mood Symptoms of Parkinson’s Disease study, were reported September 23 at the Fourth World Parkinson Congress in Portland, Oregon.
The MASTER-PD study was a multicenter, double-blind, sham-controlled study in patients with idiopathic PD and significant motor problems despite treatment with medications.
The results, soon to be published, confirmed that high-frequency bilateral M1 stimulation is beneficial in improving the motor symptoms of PD. However, which specific motor symptoms improve has been less clear.
Benefit “Modest”
Fourth World Parkinson Congress. Poster P33.67. Presented September 23, 2016.
Treatments For The General Public
Neurosurgery planning
Nexstim obtained United States Federal Food, Drug, and Cosmetic Act§Section 510 clearance for the assessment of the primary motor cortex for pre-procedural planning in December 2009 and for neurosurgical planning in June 2011.
Depression
In 2008, the US Food and Drug Administration authorized the use of rTMS as a treatment for depression that has not improved with other measures. A number of deep TMS have received FDA 510k clearance to market for use in adults with treatment resistant major depressive disorders. The Royal Australian and New Zealand College of Psychiatrists has endorsed rTMS for treatment resistant major depressive disorder .
Migraine
The use of single-pulse TMS was approved by the FDA for treatment of migraines in December 2013. It is approved as a Class II medical device under the de novo pathway.
Other neurological areas
In the European Economic Area, various versions of Deep TMS H-coils have CE marking for Alzheimers disease,autism,bipolar disorder,epilepsy, chronic pain,major depressive disorder,Parkinsons disease,posttraumatic stress disorder ,schizophrenia and to aid smoking cessation. One review found tentative benefit for cognitive enhancement in healthy people.
In August 2018, the US Food and Drug Administration authorized the use of TMS in the treatment of obsessive-compulsive disorder .
How Is Parkinson’s Disease Treated
As cognitive impairment is often present in Parkinson’s disease, screening is usually involved in clinical practice. The Montreal Cognitive Assessment is a rapid screening instrument for mild cognitive dysfunction, used to assess executive functions, memory, language, calculation and orientation, attention and concentration, and visuoconstructional skills.
Read Also: Big Exercises For Parkinson’s Disease
Whats Next For Tms And You
If youre a Parkinsons patient, the biggest question on your mind is whether transcranial magnetic therapy is right for you. Every patient is different, and what might work best for one patient might not work for you. Then again, it might. If you feel that your ongoing course of therapy isnt working, or your doctor, neurologist, or other healthcare provider has brought it up, you should bring up TMS therapy as a possibility. They may suggest a new course of medication or other therapy first, but it never hurts to ask about it. If nothing else, youll both be aware that there are options and alternatives that may provide a more satisfying and longer lasting period of reduced symptoms and diminished or eliminated side effects.
Dont be shy about bringing up TMS therapy for Parkinsons at your next doctor or neurologist visit. Your treatment is a working arrangement with your physicians, and you should all be willing to collaborate and share information during its course!
Tms Treatment For Parkinsons In 2019
In this video, we introduce Robert D. McMullen, MD who is a psychiatrist in NYC who has been performing psychopharmacology for over 30 years, the last 10 of which includes transcranial magnetic stimulation. Today, he will discuss TMS as a treatment for Parkinsons disease.
TMS has been used as a treatment for Parkinsons disease for only 10 years, however, there have already been some breakthroughs. One case that Dr. McMullen highlights is a religious lady who came to him because she had developed severe Parkinsons. She had many of the common symptoms associated with Parkinsons including slurred speech, shaky hands, and she also required assistance to walk. She had come to Dr. McMullen because she could not go any higher on the Parkinsons medication dosage without feeling sick. She had also developed depression due to not being able to spend time with her family during Passover due to her illness. After only 5 to 6 TMS treatments, her condition began to improve dramatically so much so that she had begun to walk and talk normally again.
All in all, she had undergone a total of 20 treatments and her Unified Parkinsons Disease Rating Scale had gone down from a score of 70 at the beginning of treatment to a score of 30.
Recommended Reading: Meds For Parkinson’s Psychosis
Brain Regions In Associative And Limbic Basal Ganglia Loop
Voxel based morphometry reveals increased gray matter volume of the bilateral IFC in dyskinetic patients . Right IFC engages in suppressing an already initiated manual response . One study further revealed dyskinetic PD patients have a weaker inhibitory interaction between the right IFC and contralateral MC . This finding conforms with beneficial effects of inhibitory cTBS over right IFC on LID . Another study revealed that connectivity of the right IFC with the left MC was decreased in patients with LID . Nevertheless, inhibitory cTBS over right IFC improved LID symptoms in this study as well . Authors speculated that the increased communication between the right IFC and the putamen observed in this study in patients with LID might interfere with the motor inhibition network .
Task-based functional magnetic resonance imaging revealed activation of pre-SMA after intake of levodopa in LID patients . The pre-SMA has been implicated in both the suppression and initiation of movements . This might partly explain the contradictory outcomes of two LF-rTMS studies over pre-SMA on LID .
Activation of the dorsolateral prefrontal cortex was also observed in PD patients with LID . However, it was bewildering that HF-rTMS Stimulation of the left DLPFC induced a significant MC depression . Moreover, such MC depression did not reach a significant reduction of LID symptoms .
Medicines Used In Parkinson’s Disease
The most common drug used in the management of Parkinson’s disease is Levodopa. Levodopa belongs to a class of medicines known as central nervous system agents and works by undergoing a conversion into dopamine in the brain.
The long-term use of Levodopa causes people with Parkinson’s disease to change from having no ability to move muscles, to a hyperkinetic state. This state is called Levodopa-induced dyskinesia .
Dyskinesia, as a drug side effect developed after long-term use of Levodopa, can manifest as involuntary, irregular, unpredictable muscle movements, and slow repetitive movements or abnormal postures. There are limited therapeutic options for these side effects.
Amantadine is a medicine regarded as the most effective for treating LID, but its use can cause hallucinations and its discontinuation often causes a worsening of dyskinesia.
Recommended Reading: How Does Parkinson’s Disease Begin
Rtms Study: Clinical Efficiency
A more detailed description of the clinical efficiency of rTMS can be found in our previous publication. Briefly, the patients were assessed at baseline , after the 5th and 10th sessions, and then 2 weeks and 4 weeks after the last session. As shown in Table , with a 4 weeks follow-up, our results revealed significant interaction effects between group and time in the FOGQ , MDS-UPDRS III and several gait variables . Post-hoc analyses showed that in the verum group, FOGQ score was significantly decreased at T2 and T4, while MDS-UPDRS III and gait variables were significantly improved at T1, T2, T3, and T4. However, no significant improvements were found in the sham group. Regarding the adverse effects, three subjects in the verum group and one subject in the sham group reported mild but tolerable headache.
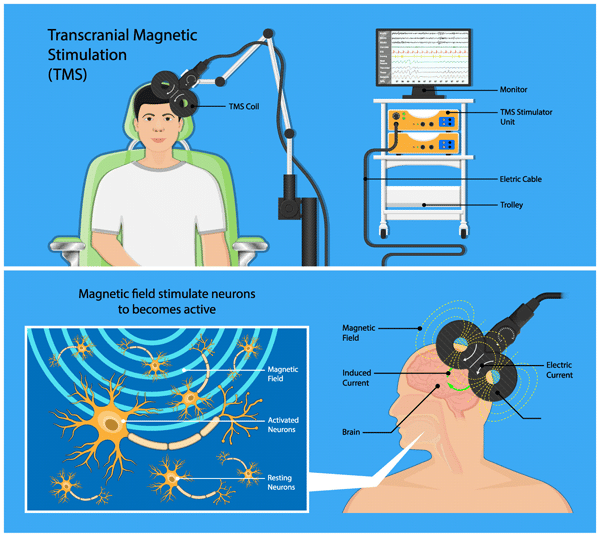
How Does Tms Treatment Work
TMS is a non-invasive method of stimulating the brain through electromagnetic pulses to the brain from a magnetic field generator coil that uses magnetic pulses similar to MRI machines. Most patients receive TMS five days a week over the course of 36 sessions. TMS sessions usually last 20-30 minutes.
You May Like: How Parkinson’s Disease Affects People
Neurotransmitter And Receptor Modulation
Imbalanced neurotransmitters are the major pathological mechanisms for LID. Studies have explored the roles of neurotransmitters and their receptors in TMS. Elevated GABA receptor levels have been found in postmortem samples of LID patients . The N-methyl-D-aspartate receptor antagonist, dextrorphan hydrochloride, has been shown to improve LID clinical outcomes . HF-rTMS increases the expression of amino acids and dopamine in the hypothalamic paraventricular nucleus and dorsal hippocampus, respectively, and decreases expression of arginine vasopressin in the hypothalamic paraventricular nucleus in healthy brains of rats and mice . LF-rTMS was, however, not capable of exhibiting any changes in neurotransmitters . Both LF-rTMS and HF-rTMS bring about an imbalance between glutamate and glutamine in healthy human brains. It has been shown that cTBS decreases vesicular glutamate transporters one and increases plasmatic glutamate transporters one in healthy rat brains . Upregulation of glutamate transporter and GABA transporter mRNAs have been reported in TMS-treated mice . Studies have confirmed the pivotal roles of glutamatergic and GABAergic neurotransmitters during TBS . More studies are needed to evaluate the role of neurotransmitters and receptors in LID patients during TMS therapy .
What Is Transcranial Magnetic Stimulation
In short, it is a non-invasive neurophysiological technique that can stimulate the human brain through the intact skull, without producing significant discomfort. In another era, we would call it a miracle. However, the lack of knowledge about its exact mechanisms takes us a bit further away from this eponymous.
It was discovered and developed by Barker and colleagues in 19853 and has been used in healthy and psychiatrically disturbed patients to explore their cortical functions. Some studies have tried to demonstrate an early therapeutic effect. Most of these studies focused on the primary motor cortex, although much attention was also diverted to depression, where it appears to be quite effective.4
Simply put, TMS uses magnetic stimulators that consist of small capacitors that discharge currents of extremely short duration through an induction coil placed on the scalp5. The idea is to create a magnetic field from the current that passes through the brain perpendicularly and reaches values up to 3 Tesla. In this process, part of the current is displaced and creates a second side in the brain, which also has some effect.
Due to the short duration of the original current, the intensity of the magnetic field decreases rapidly and does not allow it to reach long distances. Therefore, the effect is thought to be limited to the cortex and part of the subcortical white matter.6
Dont Miss: What Are The Initial Symptoms Of Parkinsons Disease
Don’t Miss: What Is The Best Medicine For Parkinson Disease
Extraction Of The Outcome Measures
The data were collected using a semi-structured form for each study by one of the authors and checked by another investigator. Discrepancies were resolved by consensus and a third author consulted if necessary. The following variables were extracted: mean and SD of the motor section of the UPDRS for baseline and after treatment for the active and placebo group mean and SD for the follow up period evaluation study design demographic and clinical characteristics baseline motor UPDRS and Hoehn and Yahr stage TMS parameters and ECT parameters .
For the studies with more than one active group , we considered each group as one study in the quantitative analysis. This approach was used for the following three studies: Mally et al , de Groot et al and Lefaucheur et al .
Research On Tms As An Alternative Treatment For Parkinsons
Some researchers hypothesize that since TMS is so effective neurologically, there is a chance that the pulses could aid the parts of the brain that control movement, which could greatly improve PD patients quality of life.
Most studies use groups of patients with PD and measure control groups who receive no treatment, several who are administered only antidepressant medication, and groups who receive TMS.
The results of these experiments reveal that while those PD patients who receive TMS treatment show great improvements to their mental health, there is little to no evidence that the neurostimulation improves motor function.
While it may not relieve the physical effects of PD, several studies concluded promising findings that this connection could exist. A meta-analysis was conducted on all studies that included patients with PD and depression. The analysis included studies that reported pre-and-post-rTMS depression inventory scores. In the findings, it was shown that out of the 7 studies, 6 showed that patients had improvement in their depression.
Another study tested 46 patients with PD and mild to moderate depression with half of the patients receiving rTMS treatment and the other half receiving a placebo. Over 10 days, patients received high-frequency rTMS applied over the primary motor cortex bilaterally. Results found that patients that actually received the treatment saw improvement in their depression and health-related quality of life.
Read Also: Can Parkinson’s Come On Quickly
Addressing Motor And Non
As discussed before, Parkinson’s disease has both motor and non-motor features. While motor symptoms can sometimes respond to therapies like deep brain stimulation, a procedure that involves implanted electrodes, non-motor symptoms usually don’t respond to it.
One non-motor symptom of Parkinson’s disease is very common. About 40% to 60% of patients undergo depression, and usually, depression in Parkinson’s disease is resistant to medication.
That is why TMS is a great potential solution. The non-invasive stimulation of brain networks makes TMS ideal to treat Parkinson’s disease, and patients have seen significant differences in motor outcomes, as well as significant improvement in non-motor outcomes. With the potential to improve both motor and mood symptoms, repetitive transcranial magnetic stimulation targets areas of the brain where neurons are underactive.
How Does Tms For Parkinsons Help
Research into using TMS therapy as part of Parkinsons treatment has shown improved motor movements when combined with regular aerobic exercise. Other research has indicated that by stimulating two regions of the brains premotor associative cortex, instead of just one, it has resulted in reduced axial symptoms associated with Parkinsons.
The studies indicate that TMS for Parkinsons treatments helps prolong the cortical silent period. The period of improvement lasted for approximately one month in the research study participants. As such, patients who receive ongoing TMS therapy, when combined with aerobic exercise, could notice marked improvements that last much longer.
Read Also: Can Yoga Help Parkinson’s Disease
Treating Comorbid Depression In Parkinsons Disease
Some estimates say half of Parkinsons disease patients suffer from clinical depression. This is often treated with antidepressant medications, but TMS could be an excellent alternative because:
- It has none of the side effects associated with antidepressants, such as:
TMS is not yet FDA-approved for treating Parkinsons, but it does have full approval for depression treatment. This means if you have a major depression diagnosis to go along with Parkinsons, insurance is likely to cover TMS .
Success TMS is a nationwide provider of transcranial magnetic stimulation with fantastic reviews.
To find out if TMS is right for you, visit SuccessTMS.com, or call at .
Were happy to help you with insurance approval and get you started on treatments as soon as youre ready.
How Does Tms Work
While there is absolutely no delivery of electric shocks or the like, it might be simpler to think of TMS as a jumpstart for the brain. As mentioned, TMS delivers magnetic pulses through a device attached to the head. Therapy can take place for about a half-hour and must be conducted over the course of several weeks to ensure it has an effect. That said, the pulses stimulate parts of the brain that become inactive due to Parkinsons or other conditions. This activates the synapses and cells that deliver neurotransmitters, chemicals that enable one cell to tell another what to do. Some have stated that TMS can help regrow these connections and revitalize the brain.
One big benefit of TMS is that its noninvasive. It doesnt require surgery or opening up the body to apply probes or the like. Also, TMS causes no pain or discomfortthough some patients report itchiness or tinglingand, again, uses no radiation or electrical shocks. With TMS, signals that might have been blocked or remained undelivered are able to get through and alleviate the symptoms affecting the bodys motion. This can open greater possibilities to keep the body active and healthy and able to fight the progression of Parkinsons disease.
Read Also: What Is The Most Common Medication For Parkinson’s Disease
Studies Utilizing Theta Burst Stimulation
Unlike rTMS, the protocol of TBS is comparatively more consistent among studies . For all five studies utilizing continuous TBS , cTBS consists of three-pulse bursts at 50 Hz repeated every 200 ms for 40 s and was administered after levodopa intake.
In Koch’s study, they firstly applied single-session cTBS on 10 PD patients with LID over the cerebellum, and a 45-min reduction was observed . In this study, a 10-day course of cTBS was further conducted and induced persistent clinical beneficial effects up to 4 weeks . However, a later study applied a 5-day course of cTBS on 8 PD patients with LID over the cerebellum only reduced LID up to 45 min .
A study applied single-session cTBS over the inferior frontal cortex and MC on 8 PD patients with LID, respectively . Stimulation over the right IFC induced improvement of LID only up to 30 min, while stimulation over MC did not exhibit any change . Although efficacy duration was not mentioned, Ponza et al. also observed the beneficial effect of cTBS on LID symptoms after single-session stimulation over the right IFC . A recent study targeting cerebellum also displayed 60-min alleviation for LID after cTBS stimulation .
Like LF-rTMS, the short-term benefits of cTBS have been corroborated in several studies and are patient-tolerable. Although a remarkably longer after effect of cTBS than of LF-rTMS was exhibited only in one study, such prolonged effect did not replicate in other studies.
