Disturbance In Sphingolipid Homeostasis And
Enhanced activation of phosphocholine cytidyltransferase resulting in increased synthesis of phosphatidylcholine as major component of phospholipid cell membranes was reported in GD . Interestingly, alterations in the lipid bilayer composition of membranes cause impaired -synuclein membrane binding and enhance aggregation-prone fibril formation . Combined 1H and 31P magnetic resonance spectroscopic imaging revealed that PDGBA patients display a disturbed membrane phospholipid metabolism in the putamen and midbrain with reduced levels of the precursor choline and increased levels of the membrane-related phospholipid degradation product glycerophosphoethanolamine. These changes were accompanied by neuronal loss in these brain regions as measured by reduced levels of the neuronal marker N-acetyl-aspartate .
An Update On Gene Therapy Approaches For Parkinsons Disease: Restoration Of Dopaminergic Function
Issue title: Repairing the Parkinsonian Brain
Guest editors: Anders Björklund, Bastiaan R. Bloem, Patrik Brundin and Howard Federoff
Article type: Review Article
Authors: Van Laar, Amber D.ab | Van Laar, Victor S.c | San Sebastian, Waldyae | Merola, Aristided | Elder, J. Bradleyc | Lonser, Russell R.c | Bankiewicz, Krystof S.ce*
Affiliations: Asklepios BioPharmaceutical, Inc., Columbus, OH, USA | Department of Neurology, University of Pittsburgh Medical Center, Pittsburgh, PA, USA | Department of Neurological Surgery, College of Medicine, The Ohio State University, Columbus, OH, USA | Department of Neurology, College of Medicine, The Ohio State University, Columbus, OH, USA | Department of Neurological Surgery, University of California San Francisco, San Francisco, CA, USA
Correspondence: Correspondence to: Krystof S. Bankiewicz, The Ohio State University, Department of Neurological Surgery, N1018 Doan Hall, 410 W. 10th Ave., Columbus, OH 43210, USA. E-mail: .
Keywords: Gene therapy, Parkinsons disease, aromatic-L-amino-acid decarboxylase, glial cell line-derived neurotrophic factor, image-guided convection-enhanced delivery, clinical trial design
DOI: 10.3233/JPD-212724
Journal: Journal of Parkinsons Disease, vol. 11, no. s2, pp. S173-S182, 2021
Abstract
Read Also: Vascular Parkinsonism And Cognitive Impairment
The Future Of Treatments For Gaucher Disease And Parkinsonism
Today, Gaucher treatments and Parkinsons treatments dont overlap. Parkinsons disease affects the brain, and Parkinsons disease treatments must cross the blood-brain barrier .
The BBB is a network of cells and blood vessels designed to keep many substances from reaching the brain. Its existence protects the brain from many types of bacteria and other foreign matter. But it makes some therapies hard to deliver.
Current Gaucher disease treatments, including enzyme replacement therapy and substrate reduction therapy, dont cross the BBB. They dont affect Parkinsons. For the same reason, they dont affect the neuronopathic symptoms of Gaucher disease types 2 and 3.
Some good news, Sidransky says, is that the link between these two disorders means that scientists are paying a lot of attention to the mechanisms that cause both conditions. That attention will lead to new therapies, including for the rarer neurologic forms of Gaucher disease.
Future treatments might involve:
Recommended Reading: When Was Muhammad Ali Diagnosed With Parkinson’s Disease
Misfolded Proteins In Gaucher Disease
When you have mutations in the GBA gene, often times the GCase protein is misfolded and never gets to the lysosome, Sidransky explains. The lysosomes function is to break down unneeded materials. In Gaucher disease, people dont have enough of the GCase enzyme, and lipids accumulate. As a result, lysosomes dont function as well as they should. Sidransky says, Some describe it as a vicious cycle: When the enzyme isnt made, lipids accumulate. The lipids block other things, which contributes to the dysfunction of the lysosomes.
A Busy Year Ahead For Parkinsons Disease
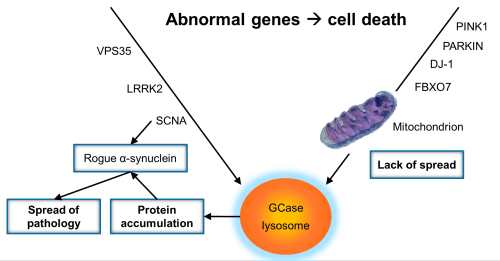
Parkinsons disease research has ended in numerous dead ends despite substantial efforts over many years. Recently, Biogen and Sanofi scrapped their Parkinsons candidates, cipanemab and venglustat respectively, owing to lack of efficacy, and a disease-modifying therapy has yet to materialise.
But the push to find drugs that help beyond reducing symptoms continues, and Evaluate Vantage has delved into the pipeline of projects in active late-stage clinical trials. This year is shaping up to be crucial for the field, with 10 studies expected to yield data or to complete in 2021.
One target that crops up multiple times is GLP-1 this approach, traditionally employed in type 2 diabetes, is also being tested in Alzheimers. Among other avenues of research, it is hoped that gene therapy could offer a one-time cure for Parkinsons.
Repurposing
Research has suggested that GLP-1 agonists have neuroprotective benefits, and several trials of marketed diabetes drugs, as well as new GLP-1-targeting projects, are under way in Parkinsons. Some of these studies are investigator sponsored, including the most advanced, a UCL-run phase III trial of Astrazenecas Bydureon called Exenatide-PD3.
In the meantime, data are expected from several phase II studies of GLP-1 agonists, including a trial of Novo Nordisks Victoza, being run by Cedars-Sinai Medical in collaboration with the Danish company and The Cure Parkinsons Trust. That study is set to complete in September.
Also Check: How To Diagnose Parkinson’s On Mri
Glucocerebrosidase Mutations Cause Mitochondrial And Lysosomal Dysfunction In Parkinsons Disease: Pathogenesis And Therapeutic Implications
- 1Department of Neurology, Peking University Third Hospital, Beijing, China
- 2Beijing Key Laboratory of Biomarker and Translational Research in Neurodegenerative Diseases, Beijing, China
- 3Key Laboratory for Neuroscience, National Health Commission/Ministry of Education, Peking University, Beijing, China
Proposed Pathogenesis Of Gba
Recent studies have been exploring the contribution of mutant GBA to PD pathogenesis. To date, the mechanisms that underlie the increased risk of developing PD among GBA mutation carriers have not been fully elucidated. In general, in autosomal dominant forms of PD, such as those involving LRRK2 and -synuclein, gain-of-function mutations are usually involved in PD pathogenesis. In contrast, loss-of-function mutations, such as those in such as parkin, DJ-1 and PINK1, are implicated in most autosomal recessive forms of PD.
Notably, the inheritance of GBA-associated PD does not follow strict Mendelian law, although GD is an autosomal recessive disease. Therefore, both gain-of-function and loss-of-function have been proposed as explanations for GBA mutation increasing the risk of PD development .
Figure 1. The schematic diagram of proposed mechanisms by which GBA mutations contribute to the development of PD. Contributions to GBA-PD pathogenesis may include enhanced -synuclein aggregation, altered lipid homeostasis, autophagy-lysosomal dysfunction and mitochondrial dysfunction. GBA, glucocerebrosidase GCase, glucocerebrosidase.
Also Check: What Medications Should Parkinson’s Patients Avoid
Who Might Be Useful For Gene Therapy And Are There Any Risks
Researchers believe that gene therapy will be useful for a number of people with Parkinsons, irrespective of whether their condition has been genetically caused. However, as with most treatments, it wont be suitable for everyone. As part of the clinical trial process, scientists will establish who this treatment will suit and those for whom it is not recommended.
As with all treatments, there are some risks. It is thought that there may be side effects within the central nervous system that relate to long-term, high levels of exposure to therapeutic genes, or there may be an immune response to the treatment. Current research efforts are directed at implementing further measures to reduce the probability of these risks, even if they are unlikely.
What Is The Future Of Gene Therapy For Parkinsons Disease
The jury is still out on each of the gene therapy strategies discussed above and each is still under investigation in an active clinical trial, although as of publication of this blog, none are currently recruiting participants. Three of APDAs Centers for Advanced Research are participating in gene therapy trials for Parkinsons disease. Emory University School of Medicine and the University of Alabama at Birmingham School of Medicine are participating in trials of Neurturin. The University of Pittsburgh Medical Center and Emory University School of Medicine are participating in trials of AADC.
Don’t Miss: What Kind Of Pain Is Associated With Parkinson’s
The Future Of Research In Neurodegenerative Disease
Additional studies across the country are now using this new technique for other neurodegenerative diseases and neurologic disorders. The strategy allows researchers to evaluate the true extent of therapeutic delivery, eliminating the possibility that an effective therapy wont work simply because it missed its mark.
Mass General has a history of making safe but innovative advances in functional neurosurgery, and were well-positioned to be an international center of excellence for gene therapy in the brain, Dr. Richardson says. We have a lot of collaborators in other areas of neurology, psychiatry and the basic sciences with whom we can partner in the future to expand the application of this technique.
Inquiries into this phase II clinical trial at Mass General can be sent to: , or call 617-726-2937.
Refer a patient to the Department of Neurosurgery
Contributors
National Institute on Aging, National Institutes of Health , United States
Reviewed by
Northwestern University, United States
The editor and reviewers affiliations are the latest provided on their Loop research profiles and may not reflect their situation at the time of review.
Progress In The Treatment Of Parkinsons Disease
Despite the fact that 200 years passed since the discovery of PD, it was not until later in the 20th century that progress in the treatment of PD was achieved, predominantly due to the limited understanding of PD pathophysiology. Given Carlssons discoveries of DAs involvement in the 1950s, it became clear that PD development involved dopaminergic cell death and a decrease of DA in the striatum and other structures of the forebrain. The first steps towards treatment were made by Carlsson , who proposed targeting this DA deficiency to facilitate symptom reduction.
Read Also: What Is Good For Parkinson’s Disease
Gba Variants Predispose To Accelerated
Post-mortem studies show enhanced aggregation and propagation of -synuclein not only in the substantia nigra and putamen but also wide-spread neocortical Lewy-body pathology in brain tissue of PDGBA and DLBGBA .
The accelerated cognitive decline PDGBA makes this subgroup of PD a good model to study CSF profiles that are associated with cognitive impairment. In general, limbic and/or cortical Lewy-body pathology is hypothesized to be the main substrate forcing driving cognitive decline in PD . In more recent years, it became clear that a considerable proportion of PD patients who developed dementia in their disease course show concomitant amyloid-beta and tau pathology at autopsy in addition to the typical Lewy-body pathology . Correspondingly, reduced CSF levels of Amyloid-beta1-42 and/or elevated CSF levels of total-Tau and phospho-Tau have been reported to be associated with cognitive impairment in PD . However, this seems not to be the case in PDGBA as CSF levels of A1-42, t-Tau, and p-Tau are similar to those seen in healthy control individuals. In light of the CSF profiles of reduced total levels of -synuclein and the prominent -synuclein seeding activity, the pronounced cognitive decline in PDGBA is driven by -synuclein aggregation and cortical Lewy-body pathology.
Parkinson’s Disease With Gba1 Mutations
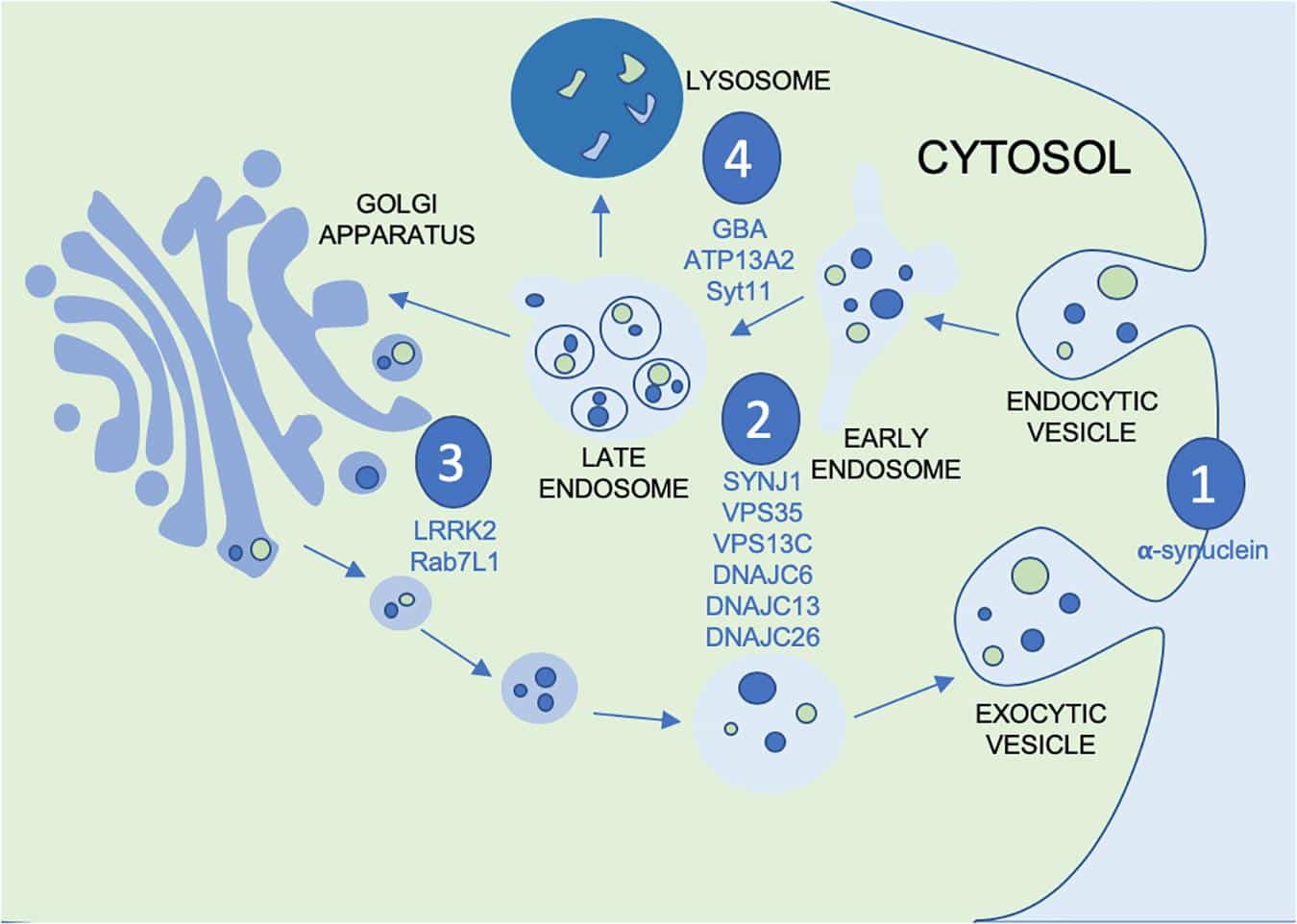
Parkinsons disease is a severe and progressive neurodegenerative disorder that affects more than seven million people worldwide and up to one million people in the United States. Although Parkinsons has historically been characterized as a movement disorder, patients can suffer from a range of non-motor symptoms, including psychosis, dementia and cognitive impairment. Pathologically, Parkinsons disease is characterized by the presence of abnormal clumps of a protein called -Synuclein that form in neurons throughout the brain. These protein aggregates are known as Lewy bodies.
There are currently no approved therapies that modify the course of Parkinsons disease or the underlying pathological process.
Large-scale genetic studies have recently identified dozens of causative and risk genes for Parkinsons disease. Many of these genes are involved in the normal functioning of lysosomes so-called recycling centers in cells that contain enzymes responsible for degrading proteins, lipids and sugars to regulate metabolic function.
In addition to Parkinson’s disease with GBA1 mutations, we are developing PR001 for the treatment of Type 1 Gaucher disease as well as Type 2 Gaucher disease.
Recommended Reading: How Often Does Parkinson’s Disease Occur In The Population
Increased Risk Of Parkinsonism In The Gaucher Community
Unfortunately, people with GBA mutations that cause Gaucher disease do have a higher risk of developing Parkinsons disease. Still, Sidransky points out that most peopleincluding those with Gaucher diseasenever develop Parkinsons.
In the general population, a rough estimate is that Parkinsons affects around 1% of the population over age 60. Small numbers of people receive a diagnosis at a younger age. Up to 5% of individuals in the general population may develop Parkinsons disease or related conditions by age 85.
People with a Gaucher gene mutation have:
- Higher frequency of Parkinsons disease: As many as 9% of people with Gaucher disease may develop Parkinsons disease. The risk is up to 3% in Gaucher carriers.
- Earlier onset of Parkinsons disease: Parkinsons disease may start earlier in people with the GBA gene mutation than in non-carriers. On average it may appear 611 years earlier in people with Gaucher and 36 years earlier in carriers, but there is a range.
- Greater likelihood of DLB: DLB cases may appear around 5 years earlier in Gaucher carriers and be more severe. This disorder is more likely in men than in women.
- More frequent RBDs:GBA mutations are also more common in people with RBDs. But people who have a GBA mutation and RBD dont appear to be more likely to progress to Parkinsons disease than those without Gaucher disease.
Secure The Cell Therapy Supply Chain From Bench To Bedside
By clicking the Download Free Whitepaper button, you accept the terms and conditions and acknowledge that your data will be used as described in the Cytiva Thematic privacy policy By downloading this Whitepaper, you acknowledge that we may share your information with our white paper partners/sponsors who may contact you directly with information on their products and services.Visit our privacy policy for more information about our services, how we may use, process and share your personal data, including information on your rights in respect of your personal data and how you can unsubscribe from future marketing communications. Our services are intended for corporate subscribers and you warrant that the email address submitted is your corporate email address.
Also Check: Can Parkinson’s Tremors Come And Go
Exploring The Genotypephenotype Correlation In Gba
- Department of Clinical and Movement Neurosciences, UCL Queen Square Institute of Neurology, London, United Kingdom
Variants in the glucocerebrosidase gene are the most common genetic risk factor for Parkinson disease . These include pathogenic variants causing Gaucher disease and risk variants, which are not associated with GD but nevertheless confer increased risk of PD. As a group, GBA-PD patients have more severe motor and nonmotor symptoms, faster disease progression, and reduced survival compared with noncarriers. However, different GBA variants impact variably on clinical phenotype. In the heterozygous state, complex and severe variants are associated with a more aggressive and rapidly progressive disease. Conversely, mild and risk variants portend a more benign course. Homozygous or compound heterozygous carriers usually display severe phenotypes, akin to heterozygous complex or severe variants carriers. This article reviews genotypephenotype correlations in GBA-PD, focusing on clinical and nonclinical aspects , and explores other disease modifiers that deserve consideration in the characterization of these patients.
Gene Therapy Transducing Gba1 In The Brain
Delivering a normal GBA1 gene using an adeno-associated virus vector to a variety of animal models of PD or PD-GBA, including various genetic models with -synuclein or GBA1 gene mutations, has been shown to be efficacious by multiple independent investigators . Most of the these in vivo studies delivered the AAV-GBA1 vector directly into the rodent CNS, although in at least two examples, intravenous delivery was used . Increasing GCase levels and activity through the delivery of a GBA1 vector has been reported to reduce inflammation as well as the accumulation of aggregated -synuclein in these multiple independent models of PD. Additional studies have used non-genetic approaches to increase GCase activity in rodent PD models and reported efficacy in the reduction of pathological features .
PR001 safety was evaluated in both mouse models and no adverse histopathologic findings or evidence of toxicity due to treatment was observed. Biodistribution and toxicology studies were performed in nonhuman primates. Broad distribution of PR001 vector and significant elevation of GCase protein levels were observed in the brain. There were no adverse findings or evidence of toxicity due to PR001.
Read Also: Stretching Exercises For Parkinson’s
Epidemiological Demographic And Prognostic Features
Compared with noncarriers, GBA-PD patients usually present symptoms earlier on . PD patients with biallelic GBA variants , hereafter referred to as GD-PD, also have an earlier age at onset compared with heterozygous carriers , indicating a possible dose effect of GBA influencing age at onset. When GBA variants were stratified, the majority of studies consistently reported earlier age at onset in severe variant carriers compared with mild or risk and in patients with null or complex alleles relative to those with missense mutations . Rarely, no differences in age at onset were found .
Motor And Nonmotor Features
Compared with noncarriers, the usual presentation of GBA-PD is that of an akinetic-rigid syndrome, with early development of motor fluctuations and dyskinesia . Stratifying by GBA variants, severe variants have been associated with more severe and rapidly progressive motor phenotype, and shorter time to development of axial symptoms such as postural instability, as opposed to mild or risk variants . At the other end of the spectrum, risk variants are more likely to associate with benign phenotypes and occurrence of motor fluctuations later in the disease course .
People with GBA-PD suffer from a higher burden of nonmotor symptoms both in the prodromal phase and during manifest disease. One study reported higher scores of the nonmotor symptoms scale in GBA-PD patients compared with noncarrier PD patients . Similar results were confirmed by another study showing that PD patients with severe variants or GD-PD patients had higher nonmotor symptoms questionnaire scores compared with PD patients carrying mild variants or noncarriers .
Increased frequency of psychiatric symptoms, such as hallucinations, delusions, and impulsivecompulsive behavior , has also been reported in GBA-PD patients vs. noncarriers . The risk of psychiatric disturbances seems to be genotype specific. Severe or complex variant carriers were more affected than mild variant carriers , and risk variant carriers showed the mildest phenotype .
Don’t Miss: Does Parkinson’s Affect Heart Rate

