Will Findings From Pd Ipsc Models Translate To Human Clinical Trials
Given the apprehensions that in vitro studies may be too artificial, human iPSC-derived neural progenitors may be transplanted into animal brains,,,. Besides ethical barriers, xenografts also raise the possibility that the healthy host tissue compensate for the impairment of the transplanted cells. Yet, if the phenotypes observed in vitro are recapitulated in vivo, pharmacological treatments could be assessed in a systemic environment, with much more realistic dosage and administration methods.
Learning From Genetic Analyses Of Pd Casecontrol Studies
We analyzed the reports from 12 international studies,,,,,,,,,,,, totaling 5650 persons living with PD in North America, Europe, and Australia. We confirmed that globally only 15% of patients report a family history of PD symptoms, while the remaining 85% of the PD population are classified as sporadic PD . However, the distinction between genetic predispositions in familial and sporadic PD is blurry. No single-gene mutation in PD has a 100% penetrance. Instead, most likely, multiple genetic risk factors act in synergy to increase the chances of both familial and sporadic PD. Such genetic susceptibilities interplay with aging and environmental factors in both familial and sporadic PD.
Fig. 2: The genomics of Parkinsonâs disease: prevalence and penetrance.
a In the world-wide population of people living with PD, ~85% of PD cases are sporadic and the remaining are familial . b Genetic mutations occur at low and varying frequencies in the PD world population . Data represented as the mean±SEM. c GWAS data suggests risk variants in fPD genes tend to be less prevalent in PD cases . d Single nucleotide polymorphisms in over 44 genomic regions show significant association to PD. Each point presents an independent SNP hit associated with PD.
Epigenomic Alterations Linked With Pd In Patient
The ability to capture unique epigenomic alterations associated with PD remains an important challenge. Reprogramming fibroblasts to iPSCs may erase age-associated and naive epigenetic signatures which could contribute to sporadic PD pathophysiology. However, an epigenetic phenotype was reported in iPSC-derived PD patient neurons,. Neuronal lines derived from LRRK2 and sporadic patients exhibited epigenomic alterations when compared with healthy controls. Hypermethylation was prominent in gene regulatory regions associated with the downregulation of transcription factors FOXA1, NR3C1, HNF4A, and FOSL2. Interestingly, LRRK2 mutant and sporadic PD patient neurons shared similar methylation patterns, which were absent in the original donor fibroblasts. A spontaneous increase in the number of DNA strand breaks and genomic damage in PD patient-derived neurons could indirectly impact genomic regulation.
Don’t Miss: Parkinsons Double Vision
Genetics: Insights Into Etiology
Improvement in genetic analysis techniques in the 1990s led to the discovery of the first genetic cause of PD: mutations in the SNCA gene encoding -synuclein . At around the same time, -synuclein was found to be the major constituent of LB, the pathological hallmark of PD . Subsequently, multiplications of the SNCA gene have been found to cause PD with penetrance increasing with gene dosage . These discoveries brought -synuclein to center stage in the study of the pathogenesis of PD and led to the hypothesis that during different stages of the disease, -synuclein spreads in a stereotypical way within the nervous system in a prion-like fashion .
Table 2 Examples of genes associated with PD risk
Genes In Parkinson Disease

For a long time PD was seen as having only sporadic origin, without considering a genetic contribution for its complex pathogenesis. However, in 1997, Polymeropoulos et al. identified mutations responsible for the appearance of the disorder, in the gene coding for -synuclein . This finding changed the way the scientific community looks to PD and led to the beginning of genetic research of the disease.
Short after this discovery new genes were associated to PD, located in different chromosomic regions. This led to the perception of PD being a polygenic disease. Since then several other genes involved in PD development were identified as well as monogenic forms of PD and several genetic risk factors.
Highly penetrant mutations in genes like SNCA, Parkin, DJ-1,PINK1, LRRK2 and VPS35 produce rare monogenic forms of the disease. Studies have shown that incomplete penetrance of LRRK2 and GBA genes are strong risk factors for PD. Additionally, more than twenty common gene variants were associated with risk of PD development.
Monogenic forms
Understanding the monogenic forms of PD gives a broader vision concerning the genetic architecture of the disorder. Apparently, there is a correlation between genes having mutations leading to PD and the ones having risk factors .
Table 3: Genes involved in monogenic forms of PD and genetic risk loci .
Monogenic forms of PD are responsible for 30% of familial cases and 3-5% of sporadic cases of the disorder .
Recommended Reading: End Stage Parkinson Disease Life Expectancy
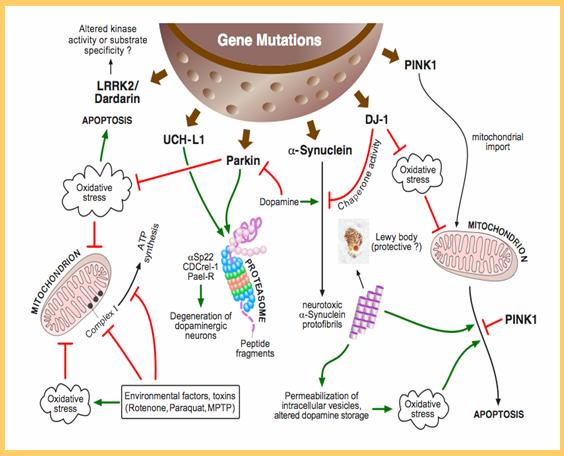
The Pink1/parkin Pathway Regulates Mitochondrial Dynamics
Mitochondrial and cytosolic GTPases playing key roles in mitochondrial fusion and fission have been previously involved in human neurodegenerative disorders, highlighting the relevance of these processes to the physiology of the cell : mutations with autosomal dominant inheritance in opa-1 , involved in fusion of the inner mitochondrial membrane, lead to retinal ganglion cell degeneration responsible for the most common form of inherited optic atrophy; autosomal dominant mutations in mfn2 , coding for a pro-fusion protein embedded in the outer mitochondrial membrane, cause the common peripheral sensorimotor neuronopathy Charcot-Marie-Tooth disease type 2A; the heterozygous A395D mutation in drp1 , encoding a cytosolic GTPase involved in mitochondrial and peroxisomal fission, has been reported to cause abnormal brain development leading to early postnatal mortality.
Genes With A Possible Role In Pd
Apart from the genes causing the six monogenic forms of PD, changes in a large number of additional genes were considered PD-causative and identified by linkage analysis or a candidate gene approach. Some of these genes even attained a PARKI designation . However, as discussed in the Genetic Classification of PD section, the link of some of these genes to PD is uncertain and not confirmed. Furthermore, mutations in some PARKs cause PD that is an inconsistent or only a minor feature of a more complex phenotype or are a very rare cause of PD . In addition, mutations in synphilin-1, NR4A2/Nurr1, POLG, mortalin, and recently presenilin-associated rhomboid-like protein were considered pathogenic based on the known function or expression/protein interaction pattern of the proteins they encode. Nevertheless, they too, are now recognized as only a minor contributor to the pool of genetic PD if at all.
You May Like: Is Gabapentin Used For Parkinson’s Disease
Monogenic Variants Of Parkinson Disease
Disease phenotypes associated with the PARK1–9 chromosomal loci follow a typical MENDELIAN pattern of inheritance , whereas PARK10 and PARK11 represent susceptibility loci with as yet undefined modes of transmission. In Mendelian genetics, the relationship between genotype and phenotype is not always readily apparent; for example, as illustrated in Figure 2, single heterozygous mutations in ‘recessive’ genes can act as susceptibility factors, thereby appearing pseudodominant. Similarly, dominant forms can present in a pseudorecessive fashion, and heritability should be suspected even in early-onset patients with a negative family history . An apparent lack of heritability might be explained by small family size, nonpaternity, adoption, variable clinical characteristics, reduced PENETRANCE, or de-novo mutations. Conversely, because sporadic PD is a relatively common condition, familial PD might be phenocopied by an occurrence of sporadic PD in a pedigree with a well-established genetic background of the disease.
Figure 2.
Recessively Inherited Parkinson Disease: Probable Loss-of-Function Mechanism
Recently, two biochemical modifications of Parkin were identified in cellular studies and human brain specimens. These data indicated that reduced E3-ligase activity of the wild-type Parkin protein could also occur as a result of the principal pathogenetic process that is responsible for the development of sporadic PD.
Figure 3.
Parkinson Disease : A Progressive Disorder That Affects The Nervous System
Parkinson disease is known as a progressive disorder that affects the nervous system. Some of the main symptoms of the disorder include tremor, muscular rigidity and slow imprecise movement. On the other hand Alzheimer âs disease is the most common cause of dementia. The disorder includes memory loss along with difficulties with thinking, problem-solving or language.Both Alzheimerâs and Parkinsonâs have many similarities but evidence may suggest they are separate disorders. Both Parkinsonâs and Alzheimerâs
Read Also: Does Parkinson’s Cause Swelling
Ii Genetic Bases Of Parkinson’s Disease
Our understanding of the mechanisms underlying the initiation and progression of PD began with the identification of mutations in the gene encoding -synuclein and the demonstration that -synuclein is the major component of filamentous Lewy bodies . Since then, at least 16 loci and 11 genes have been associated with inherited forms of parkinsonism , including PARK1 and PARK4/SNCA, PARK2/parkin, PARK5/ubiquitin COOH-terminal hydrolase L1 , PARK6/PTEN-induced kinase 1 , PARK7/DJ-1, PARK8/Leucine-rich repeat kinase 2 , PARK9/ATPase type 13A2 , PARK11/Grb10-interacting GYF protein 2 , PARK13/Omi/Htra2 , PARK14/phospholipase A2 group VI , and PARK15/F-box protein 7 . The causal genes at four genetic loci have not yet been nominated . A recent genome-wide linkage screen of 6,000 single nucleotide polymorphisms in 278 families of European descent identified two novel loci on chromosomes 3 and 18 .
Table 1 Summary of Parkinson’s disease-associated loci and genes
| PARK Loci | |
|---|---|
| Unclear | Heterozygous GD-associated mutations increase risk for PD |
PD, Parkinson’s disease; GD, Gaucher’s disease; SCA2, spinocerebellar ataxia type 2; KRS, Kufor Rakeb syndrome; SNCA, -synuclein; PINK1, PTEN-induced kinase 1; LRRK2, leucine-rich repeat kinase 2; SPR, sepiapterin reductase; UCHL1, ubiquitin COOH-terminal hydrolase 1; GIGYF2, GRB10-interacting GYF protein 2; PLA2G6, group VI phospholipase A2; GBA, -glucocerebrosidase; SNP, single polymorphism nucleotide.
Causal And Associated Genes
The idea that a gene abnormality may cause some cases of Parkinsons dates back to 1997. At that time, scientists at the National Human Genome Research Institute ;and the National Institutes of Health ;first precisely identified;that an irregularity in the;synuclein;alpha gene , the gene that provides instructions to make the protein alpha-synuclein, could lead to this movement disorder.
Alpha-synuclein is found in abundance in the brain and is thought to help regulate the release of dopamine, a chemical involved in the transmission of signals between nerve cells . With Parkinsons, the brain doesnt produce enough dopamine. This 1997 research on SNCA confirmed that at least one form of Parkinsons disease is inherited.
Up until 1997, people did not broadly think that Parkinsons could be hereditary or familial, says James Beck, PhD, chief scientific officer with the Parkinsons Foundation. With that discovery, we began to identify a number of genes linked with Parkinsons.
In 2004, scientists discovered the most common genetic contributor to Parkinsons, a mutation in LRRK2, a gene that is active in the brain and pushes a persons risk to 30 percent. Certain ethnic groups are more likely to have this gene irregularity. The faulty LRRK2 gene accounts for 1 percent to 2 percent of all Parkinsons cases, according to a review published in February 2016 in Biochemical Journal.
Recommended Reading: How Do You Find Out If You Have Parkinson Disease
What Genes Are Linked To Parkinsons Disease
In 1997, we studied a large family that came from a small town in Southern Italy in which PD was inherited from parent to child . We found the gene that caused their inherited Parkinsonâs Disease and it coded for a protein called alpha-synuclein. If one studies the brains of people with PD after they die, one can see tiny little accumulations of protein called Lewy Bodies . Research has shown that there is a large amount of alpha-synuclein protein in the Lewy Bodies of people who have non-inherited PD as well as in the brains of people who have inherited PD. This immediately told us that alpha-synuclein played an important role in all forms of PD and we are still doing a lot of research to better understand this role.
Currently, seven genes that cause some form of Parkinsonâs disease have been identified. Mutations in three known genes called SNCA , UCHL1 , and LRRK2 and another mapped gene have been reported in families with dominant inheritance. Mutations in three known genes, PARK2, PARK7 , and PINK1 have been found in affected individuals who had siblings with the condition but whose parents did not have Parkinsonâs disease . There is some research to suggest that these genes are also involved in early-onset Parkinsonâs disease or in dominantly inherited Parkinsonâs disease but it is too early yet to be certain.
E Lessons From Neuropathology
PD with autosomal recessive inheritance differs generally from idiopathic PD, although cases with a clinical course indistinguishable from that of the typical disease have been reported. Autosomal recessive PD is characterized by 1) early disease onset, in most cases before age 40; 2) benign, slowly progressive disease course; 3) excellent response to levodopa but early levodopa-induced dyskinesias; and 4) minimal cognitive decline, minimal dysautonomia. It is consistent with neurodegeneration mainly restricted to the dopaminergic neurons of the SNc, as confirmed by the neuropathological analyses of the few cases that have come to autopsy. The only autosomal recessive forms of PD that have been examined post mortem for brain pathology are parkin- and, most recently, PINK1-linked diseases.
In light of all these observations, it becomes increasingly clear that the subdivision of parkinsonian syndromes into two physiopathological groups with distinct neuropathology, Lewy body-PD-related autosomal dominant forms and recessive parkinsonism without synucleinopathy, is to a certain degree artificial and may be misleading .
Don’t Miss: How Does Dopamine Affect Parkinson’s Disease
Can Parkinsons Be Passed From Parent To Child
Its rare for Parkinsons disease to be passed down from parent to child. Most cases of Parkinsons arent hereditary. But people who get early-onset Parkinsons disease are more likely to have inherited it.
Having a family history of Parkinsons disease may increase the risk that youll get it. This means that having a parent or sibling with Parkinsons slightly increases the risk.
In most cases, the cause of Parkinsons disease remains unknown. But researchers have identified multiple risk factors that can increase your chances of getting this disease.
Risk factors for Parkinsons disease include:
- mutations in specific genes associated with Parkinsons
- having a family history of Parkinsons or a first-degree family member with Parkinsons
- being older, especially above the age of 60
- exposure to herbicides and pesticides
- being assigned male at birth
- history of brain injury
What Research Is Being Done
Current research programs funded by the National Institute of Neurological Disorders and Stroke are using animal models to study how the disease progresses and to develop new drug therapies. Scientists looking for the cause of PD continue to search for possible environmental factors, such as toxins, that may trigger the disorder, and study genetic factors to determine how defective genes play a role.; Researchers are investigating how cellular processes controlled by genes contribute to neurodegeneration , including the toxic accumulation of the protein alpha-synuclein in nerve cells and how the loss of dopamine impairs communication between nerve cells.; Other scientists are working to develop new protective drugs that can delay, prevent, or reverse the disease.; Key NINDS programs include the Parkinson’s Disease Biomarkers Program, which is aimed at discovering biomarkers–new ways to identify people at risk for developing PD and to track the progression of the disease; and the Accelerating Medicines Partnership for Parkinson’s Disease , in which the National Institutes of Health, multiple biopharmaceutical and life science communities, and nonprofit advocacy organizations work to identify and validate biomarkers for PD.; More information about Parkinson’s Disease research is available at:
The four primary symptoms of PD are:
The four primary symptoms of PD are:
The four primary symptoms of PD are:
Recommended Reading: How Does Levodopa Help Parkinson’s Disease
The Pink1/parkin Pathway Regulates Mitochondrial Quality Control
Two recent studies have provided strong evidence that the ubiquitin-protesome system plays a role beyond Mfn1/2 degradation in Parkin-dependent mitophagy : Parkin appears to activate the ubiquitin-proteasome system to promote the degradation of a series of proteins of the OMM of damaged mitochondria, leading to the rupture of the OMM, and only secondarily to the autophagic degradation of the disrupted organelles. These findings are consistent with the idea that the PINK1/parkin pathway may only be required for preparing mitochondria for mitophagy, a process termed priming and associated with ubiquitylation of the organelles, but not for induction of the process . In contrast, the BH3-only Bcl-2 family protein Nix, previously shown to be essential for elimination of mitochondria during erythroid cell maturation , appears to be required for mobilization of the autophagic machinery. Particularly, it was shown that Nix is required for membrane depolarization and generation of a burst of reactive oxygen species resulting in inhibition of mTOR signaling, mitochondrial translocation of Parkin, and induction of mitophagy .
People Who Already Have Pd: Should I Get Tested And What Do I Do With The Results
Up until recently, even people with PD with a very extensive family history of PD would not necessarily receive genetic testing because there were no clear uses for the results. There has been research directed at figuring out whether PD caused by or associated with certain mutations have particular clinical characteristics . However, there remains so much variability in clinical characteristics even among people with the same PD mutation, that there are still no clear practical implications in knowing whether a PD patient harbors a particular mutation. There is also, so far, no difference in treatment or management of PD whether or not the patient harbors one of the known mutations. That may change however, with the advent of clinical trials that target particular mutations.
There are two genes that have received particular attention recently because medications are being developed that target those with mutations of these genes.
GBAis a gene that increases the risk of developing PD. The gene encodes for the GBA enzyme, a protein used by the body to break down cellular products. Having two abnormal GBA genes causes Gaucherâs disease, which is characterized by the buildup of these cellular products resulting in fatigue, bone pain, easy bleeding and an enlarged spleen and liver. When a person inherits only one abnormal gene, he or she does not develop Gaucherâs disease, but does incur a small increased risk of PD. Most people with one mutated GBA gene do not develop PD.
Read Also: Can Mold Cause Parkinson’s Disease
