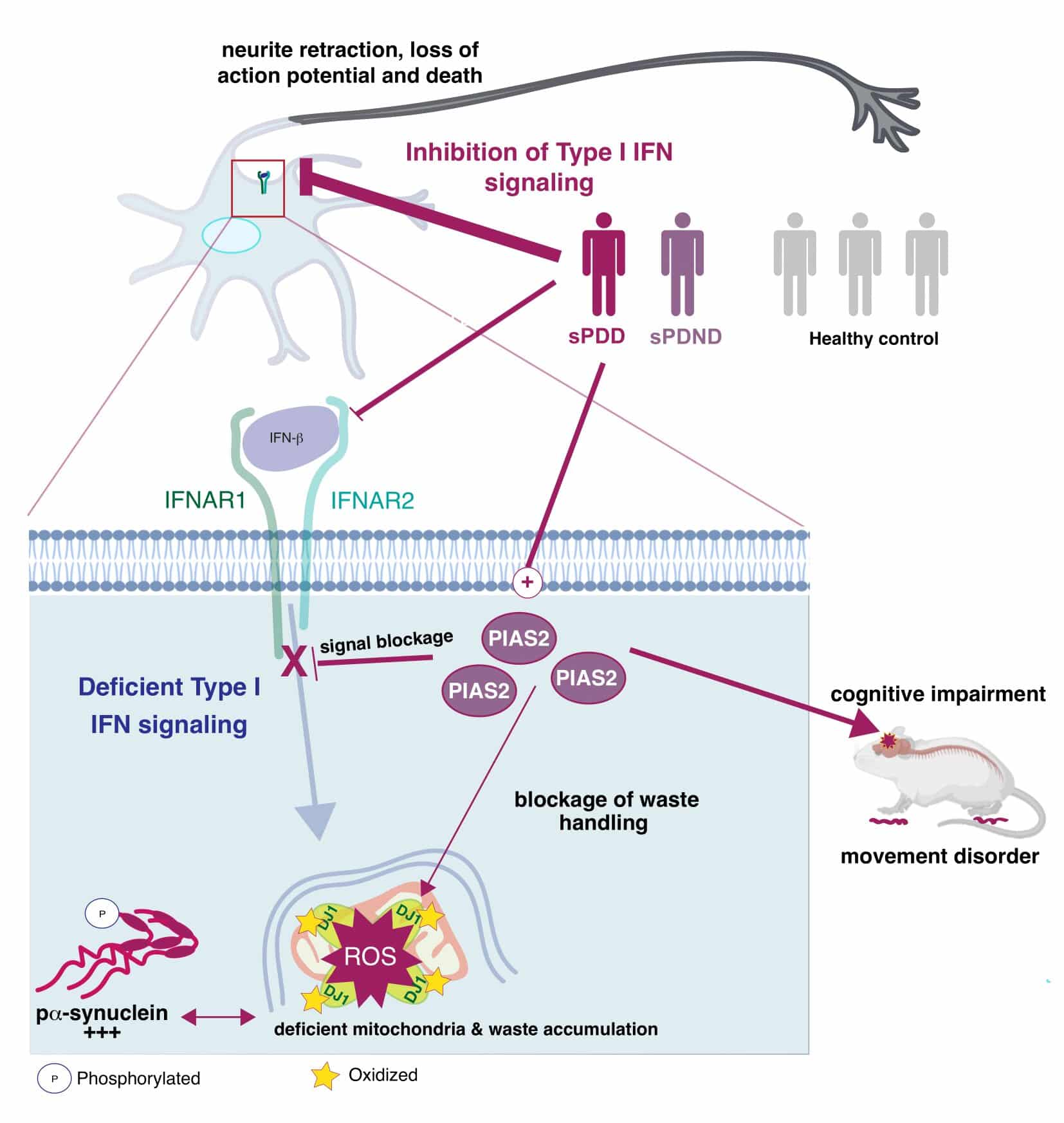
Implications For Genetic Testing And Potential Therapeutic Options To Ameliorate Mitochondrial Function In Pd
Currently, only genetic testing allows identifying patients with probable mitochondrial dysfunction by detection of variants in genes associated with mitochondrial pathways. Nevertheless, at present, only a minority of PD patients undergo genetic testing.
A variety of drugs are used in clinical practice to treat PD, mostly by increasing dopamine levels in the midbrain . However, these approaches only allow for symptomatic treatment, and no neuroprotective effect has been demonstrated with any of the drugs approved to date. Such disease-modifying treatment options are urgently needed as neurodegeneration progresses during the disease course, and symptomatic treatment is not able to prevent severe disability and a significant decrease in the quality of life in later disease stages .
Various therapeutic approaches focus on a possible mitochondrial etiology of PD: First, several approaches target the presence of ROS. Although positive effects were observed with various substances in vitro and in vivo in animal models, only the antioxidant substance MitoQ that was reported to protect dopaminergic neurons in 6-OHDA-treated mice reached the testing in clinical trials. Unfortunately, there was no evidence for neuroprotection in PD patients .
Source Of Ros Production
Mitochondria are the main source of reactive oxygen species , which are highly reactive chemical species derived from molecular oxygen. They are generated as a result of the abnormal processing within the inner membrane of mitochondria. Specifically, it happens when electrons skip a normal sequence of reaction steps and react with free oxygen. Impaired mitochondrial functions further increase the levels of ROS.
Low levels of ROS are not of concern. Infact, they participate in various physiological processes in the cell. However, when generated at abnormally higher levels, they become a threat to the cell.
Cells have evolved numerous antioxidant defense systems to counteract the negative effects of ROS. But sometimes, these defense mechanisms are insufficient to keep ROS below the toxic levels. This results in a condition called oxidative stress.
Oxidative stress is strongly linked to Parkinsons disease. It destroys the dopamine producing cells and ultimately leads to Parkinsons like changes in the brain. This is supported by numerous postmortem studies where brains from patients were found to have increased levels of oxidative stress.
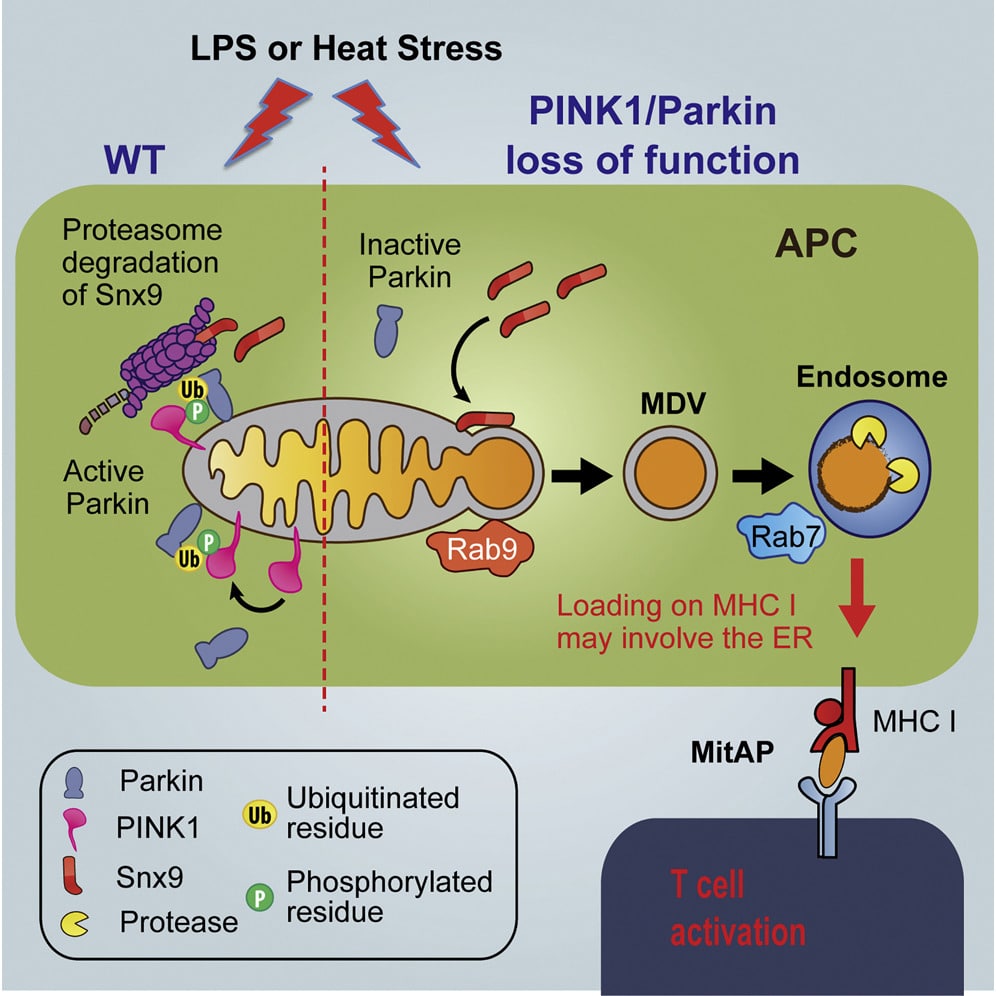
Mcu Inactivation Lends Resistance To Mptp Toxicity
mcu inactivation renders neuroprotection against MPTP. Representative images of MPTP-untreated wt , pink1/ , / , mcu/ and MPTP-treated wt , pink1/ , / and mcu/ 3dpf larvae after WISH using TH-specific riboprobe. MPTP-treated wt and pink1/ zebrafish larvae were most susceptible to MPTP toxicity while MPTP-untreated / and mcu/ were most resistant to MPTP toxicity. Graphical representation of chromogenic WISH. There was a significant decrease in number of dopaminergic neurons in pink1/ zebrafish treated with MPTP when compared to untreated pink1/ zebrafish. MPTP-treated / zebrafish showed a significant increase in number of dopaminergic neurons when compared to MPTP-treated pink1/ zebrafish. Statistical analysis with one-way anova and post hoc analysis using Tukey’s multiple comparison test of two different experiments with n=20. KaplanMeier survival curves depicting survival rate for 5 days of wt, pink1/, mcu/ and / zebrafish treated with 25 µg ml-1 MPTP . Scale bars:100µm.
Don’t Miss: What Is Parkinson’s Disease Characterized By
Mitochondrial And Proteasomal Cross
After discussing the possible involvement of mitochondria dysfunction and proteasome impairment in both familial and sporadic forms of PD, we will discuss the possible molecular mechanisms that underline mitochondrial and proteasome interplay in the disease course .
Mitochondrial and proteasomal cross-talk in PD pathogenesis. The four mitochondrial main functions are ATP production regulation of calcium homeostasis oxidative stress and apoptotic cell death mediation. Proteasome is a major protein degradation cell complex. Reduction in mitochondrial cx I activity in LOPD and the acute model of PD inhibit proteasome activity, as it actually happens in LOPD and with aging. Also in PARK6 there is mitochondrial function compromise and therefore proteasome impairment. However, recent and preliminary results shown an up-regulation of proteasomal activity in ndufa knockdown cells. In the opposite way of this two ways crosstalk, lactacystin and/or mutations in genes that code PINK1 , parkin and DJ-1 may interfere with the function of the ubiquitin E3 ligase activity complex, inducing alpha-synuclein aggregation and mitochondrial-mediated apoptosis. Accumulation of alpha-synuclein oligomers potentiates proteasome impairment in a vicious cycle. Also described in the figure are parkin function of induce autophagy in dysfunctional mitochondria and UCHL-1 deubiquitinating activity, impaired in PARK2 and PARK5, respectively.
Intervention Points For Mitophagy
PINK1-Parkin-dependent and -independent pathways provide many potential biological intervention points to enhance mitophagy. Indeed, several molecules are already available to perturb biology through inhibition or activation of a specific target . These molecules are useful to address hypothesis validity, and in some instances may provide a starting point for therapeutic development. Several additional mitophagy regulators hold potential for future therapeutic targeting.
Don’t Miss: How To Get Tested For Parkinson’s Disease
Phosphatase And Tensin Homolog
Studies involving uncoupling experiments have demonstrated that loss of mitochondrial membrane potential prevents PINK1 import to the inner mitochondrial membrane and undergoing subsequent proteolysis, which results in an accumulation of PINK1 on the surface of the mitochondria . The consequence of PINK1 accumulation on the outer mitochondrial membrane is discussed in the next section.
Mitochondrial Dysfunction Caused By Pd
The PINK1/parkin pathway is involved as well in the balance between fusion and fission. Indeed, Mfn2 is a substrate of PINK1 and Parkin. Its ubiquitination and phosphorylation lead to its degradation. Which means that upon depolarisation, PINK1 and Parkin accumulate on the MOM in their active form, Mfn2 becomes degraded and mitochondria subsequently fragmented .
Besides PINK1 and Parkin, another PD-linked protein related to juvenile forms of the disease is associated with mitochondrial quality control: DJ-1 . Loss of function mutations in the DJ-1 gene have been associated with autosomal recessive early onset PD and are far rarer than PINK1 or Parkin mutations . DJ-1 has been described as a sensor of cellular oxidative stress. When stress occurs, DJ-1 is oxidised and subsequently translocated to the mitochondria . The PD-associated loss of DJ-1 function is related to mitochondrial damage and to an increased vulnerability to complex I inhibition, as has been shown in vivo . Moreover, DJ-1 associates with molecular chaperones such as mortalin to protect cells against stress-induced apoptosis . The loss of DJ-1 function has been linked to reduced lysosomal activity and reduced basal autophagy with an accumulation of dysfunctional mitochondria in patient-based cellular models . This indicates a convergence of DJ-1-related pathogenic pathways with the PINK1/Parkin-mediated mitophagy, as the major cellular degradation pathway for dysfunctional mitochondria.
Don’t Miss: What Is The Difference Between Essential Tremor And Parkinson Disease
Does Mitochondrial Dysfunction Drive The Development Of Neurodegeneration In Pd
It is evident from other conditions, for example, mitochondrial disorders associated with mutations in the polymerase gamma gene, POLG, that a primary mitochondrial defect is sufficient to cause loss of SN neurons . Furthermore, such mutations are often associated with the development of PD-like symptoms . However, attempting to disentangle cause and effect in the contribution of mitochondrial dysfunction to the pathogenesis of PD is difficult, since many of the changes we detect in those with PD are also present in healthy aged individuals. The mitochondrial defects present in these individuals often exist in the absence of cell loss or parkinsonian symptoms. For example, equivalent levels of mtDNA deletions are detected in SN neurons from individuals with PD and healthy controls and neurons showing deficiencies for both Complex I and Complex IV are also detected in both instances . Many of the processes described above initiate changes in other pathways and thus activate well defined responses which exist to mitigate to these changes, for example, ROS and antioxidants. Therefore, it might be suggested that in PD it is the ability of neurons to respond to mitochondrial functional changes that is impaired. Thus, neurons become more sensitive to changes in other pathways, which ultimately leads to their degeneration and loss.
Relationship Between Mitochondrial Dysfunction
Considering the role of mitochondria in the pathogenesis of PD, it is necessary to evaluate the relationship between this organelle and -syn, which represents the pathological hallmark of PD. Indeed, this protein represents the principal constituent of Lewy bodies, and the point mutation A53T in its gene was the first to be identified in 1997 in hereditary PD . Alpha-syn is a 140-amino-acid long protein that is mainly located in the presynaptic terminal of neurons where it is involved in synaptic vesicle turnover and endocytosis. Particularly, it is involved into the synthesis, storage, and release of dopamine within neurons of substantia nigra .
Figure 3. Interaction of misfolded proteins and mitochondria in neurodegenerative disorders. Mitochondrial dysfunction leads to deficit in metabolic function and calcium homeostasis, apoptosis, and possibly impairment of the ability of mitochondria to degrade and import proteins. Amyloidogenic proteins such -syn and A accumulate in cytosol and aggregate, preventing also mitochondrial import of other proteins. These events increase mitochondrial dysfunction in a vicious cycle. -syn, alpha-synuclein A, amyloid beta ROS, reactive oxygen species.
Alpha-syn has also been demonstrated to interact within the interconnection between ER and mitochondria, which are essential to neuron survival . In stress conditions, ER transfers high levels of Ca2+ to the mitochondria, increasing the production of ROS .
Don’t Miss: Does Agent Orange Cause Parkinson’s
Enhancing The Clearance Of Dysfunctional Mitochondria Via Mitophagy Or Other Mitochondrial Stress Response Pathways
Multiple lines of evidence point to the importance of mitophagy in the pathophysiology of PD. For example, the PRKN and PINK1 genes mediate mitophagy and are the major causes of autosomal recessive early onset mPD . Therefore, enhancing mitophagy is a key therapeutic strategy in PD . In keeping with this, investigators used a rodent model of PD to study the effect of kinetin, the precursor of kinetin triphosphate, an activator of both wild-type and mutant forms of PINK1 . However, in PINK1 null rodents, no degeneration of midbrain dopamine neurons was identified. Additionally, in rodent models of -synuclein induced toxicity, boosting PINK1 activity with oral kinetin provided no protective effects, thus showing no evidence of a beneficial effect in a preclinical model of IPD . Another agent, celastrol, was shown to exert neuroprotective effects through activating mitophagy and inhibiting dopaminergic neuronal loss in PD cell and mouse models . Recently, a study used a high-throughput phenotype detection system for drug screening in dopaminergic neurons from induced-pluripotent stem cells derived from patients with PD due to PRKN or PINK1 mutations . After screening 320 compounds, they identified 4 candidate drugs that were effective for ameliorating impaired mitochondrial clearance, showing the utility of this method for identifying candidate PD drugs .
Biomarkers For Developing Mitophagy
Development of effective therapies for PD requires biomarkers to complement clinical assessment of patients. These biomarkers need to be clinically relevant, sensitive to therapeutic effects or underlying biology, and accessible to investigation. However, very few validated biomarkers of any kind are currently available for PD. For therapeutic trials in PD, biomarkers are needed at several stages, commencing with patient selection or stratification. Biomarkers of mitochondrial or mitophagic dysfunction are needed to identify those patients who are most likely to benefit from mitophagy enhancement therapies, improving efficiency and effectiveness of the trial. During a trial, biomarkers are needed to directly analyze target engagement, if possible, and also to measure downstream biological effects of potential therapeutics, to complement use of endpoints based on clinical measures. Finally, if specific side effects are expected from modulating mitophagy, biomarkers for these negative events are also important. Availability of biomarkers is an area of unmet need in the PD research field and much work is being done to identify and validate appropriate biomarkers, as reviewed elsewhere . This section specifically discusses biomarkers that may be useful in developing therapies targeted at improving mitophagy and mitochondrial function in PD .
Recommended Reading: How I Was Diagnosed With Parkinson’s
Restoring Mitochondrial Dynamics And Trafficking
Mitochondria form a complex network, the cohesiveness and shape of which is of direct functional relevance . Mitochondria are highly dynamic, constantly breaking off, spatially relocating, and rejoining the network. This is critical for neurons as their complex architecture requires mitochondrial relocation from the soma to dendrites, axons and synapses to meet regional metabolic demands . The majority of genetic PD proteins locating to mitochondria and mitochondria-ER contact sites are associated with processes influencing or influenced by mitochondrial dynamics and trafficking . A comprehensive summary of mitochondrial dynamics and trafficking in neuronal function can be found elsewhere .
Mitochondrial dynamics are highly complex but strictly balanced processes controlled by a diverse array of established and emerging molecular mediators, in addition to mitochondria-ER contact sites . Fragmentation of the mitochondrial network is a salient observation in PD, highlighting mitochondrial dynamics as a key therapeutic target. To this end, mdivi-1, an inhibitor of the mitochondrial fission GTPase Drp1, has been used to inhibit mitochondrial fragmentation in an -synuclein rat model of PD, reducing neurodegeneration, -synuclein aggregation, mitochondrial dysfunction and oxidative stress . While therapeutic manipulation of mitochondrial dynamics shows great promise, it will require greater understanding to be effectively exploited.
In Pd Postmortem Studies Show Multiple Markers Of Impaired Of Mitochondrial Function
Studies in parkinsonian models have also shown that dysfunction of the mitochondria permeability transition pore is caused by increased levels of the mPTP-associated protein, cyclophilin D, which enhances permeability of the mPTP to calcium . A rise in mitochondrial calcium plays a key role in cell death because it dissipates the mitochondrial membrane potential, impairing ion homeostasis and ATP synthesis . Interestingly, peptidyl-prolyl cis-trans isomerase F -null mice, which lack cyclophilin D, are greatly shielded from mitochondrial calcium overload and cell death because of oxidative stress , whereas mice over-expressing cyclophilin D are more susceptible to calcium overload and cell death . Together, these postmortem studies, in combination with mechanistic mouse model and in vitro studies show that, in patients with PD mitochondrial dysfunctional likely occurs on many levels.
Also Check: What Kind Of Doctor Diagnoses Parkinson’s
Imaging The Ap Region
Neurons of AP in the hindbrain region were selected for the experiment and image analysis. They have a relatively large diameter of about 7m and convenient localization near the skin . At 5dpf, zebrafish were paralyzed by administration of 0.6µg µl1 pancuronium bromide in E3 for 7min . The immobilized embryo was mounted in the microscopic chamber by submerging in 1.5% low melting-point agarose at 37°C and drawing the embryos into a glass capillary. Zeiss LSFM Z.1 was used for all imaging experiments. After mounting the zebrafish embryo in the microscopic chamber filled with E3 at 28°C, recording of AP region was initialized, and medium in the chamber was exchanged to E3 with 10M CCCP . The acquisition was stopped 1min after Ca2+ efflux. Imaging parameters were as follows lens Zeiss 40×, 7.5ms exposure time, 40 z-stack in 39m range, 1.6s for one-time lapse. Fish were imaged from the dorsal side of the head and were submerged in the medium from the tip of the head up to the middle of the swimming bladder.
Mitochondrial Dysfunction And Os In Pd Pathogenesis
Widely accepted pathogenic mechanisms in a subset of people with PD are aberrant mitochondrial form and function. Impaired mitochondrial function leads to increased OS and affects a number of cellular pathways, leading to damage of intracellular components and to cell death. OS is one of pathogenic mechanisms of nigral dopamine cell death in PD. The etiopathogenesis of sporadic PD is complex with variable contributions of environmental factors and genetic susceptibility. Both environmental and genetic factors influence various mitochondrial aspects, including their life cycle, bioenergetic capacity, quality control, dynamic changes of morphology and connectivity , subcellular distribution , and the regulation of cell death pathways .
Mitochondria dysfunction and dopaminergic cell death in PD pathogenesis. Multiple factors, including genetics, aging and environmental toxins, or combinations, have been implicated in the aetiology of PD. Abnormal metabolic function, abnormal morphology, and impaired fission-fusion balance have all been observed in mitochondria in at least some forms of PD. Increased OS can lead to impaired function of the UPS, thereby further affecting cell survival. All these may directly or indirectly affect the mitochondrial function of protein degradation systems, including UPS and ALP, and thereby, cause the death of dopamine neurons.
Environmental factors
Ageing
Environmental toxins
Genetic factors
Contributions of PARK PD-associated genes
Read Also: Can Parkinson’s Cause Heart Problems
Mitochondrial Dysfunction Affecting Cellular Integrity
In cases of sustained mitochondrial dysfunction, apoptosis of the cell is the last solution to avoid general damage to the organism. Apoptosis is a physiological event occurring during development and arising when molecular and organellar quality controls are overwhelmed. Two distinct pathways have been described, the extrinsic and the intrinsic pathways, with the latter involving a mitochondrial signalling pathway. Here, upon cellular stress, Bax and Bak are translocated in the mitochondrial OM where these proteins colocalise with mitochondrial fission sites . In order to trigger apoptosis, these pro-apoptotic proteins need to outbalance anti-apoptotic proteins like Bcl-2 and Bcl-xL . Bax and Bak then contribute to the release of cytochrome c through the permeability transition pore . Cytochrome c will subsequently form a complex with pro-caspase-9, which activates caspase-9, the initiator caspase. Caspase-9 will in turn promote the activation of caspase-3, the executioner caspase and lead to the activation of the apoptosis pathway.
VPS35 also has an anti-apoptotic role via its association with Lamp2a and with the Parkin substrate, aminoacyl-tRNA synthetase complex interacting multifunctional protein-2 . For VPS35 harbouring the PD-associated D620N mutation, this association was disturbed and led to an increased level of non-degraded AIMP2, which translocates to nucleus and activates PARP1 leading to cell death .
Structural Alterations In Mitochondria
Mitochondrial dynamics in the form of mitochondrial fission and fusion are a cellular response to mitochondrial stress. In this study, mitochondrial area was investigated in the dopaminergic neurons across the experimental groups. Dopaminergic neurons were marked with TH primary antibody and mitochondrial structures were marked with Tom20 antibody DAPI staining was added for contrasting background . There was a significant reduction in mitochondrial area observed in pink1/ zebrafish when compared to wt and mcu deletion partially restored mitochondrial area . Alternatively, mitochondrial volume was studied in muscle fibers using transgenic zebrafish expressing mitochondria-localized GFP and imaging was performed using LSFM . There was a significant change of mitochondrial volume in muscle fibers , however mcu deletion did not restore mitochondrial volume. Conversely, mitochondrial structure was altered in the form of increased sphericity in pink1/ zebrafish when compared to wt mcu deletion restored mitochondrial sphericity .
Recommended Reading: Can Parkinson’s Start In The Legs
