Study Procedures And Assessments
Participants visit the study site three times for data collection: an initial baseline visit and two annual follow-up visits. Data collection during each visit takes a full day and can be spread out over a 1.5-day period if desired by the patient. Prior to each visit, study participants will be offered a complimentary hotel stay, thereby reducing the need for patients living far away from the study center to travel long distances in the early morning. Patients arrive at the study site in the morning in a practically defined OFF state, i.e., at least 12h after having taken their last dopaminergic PD medication.
Table 1 Overview of included study measures and scales in the Personalized Parkinson Project
After each visit participants complete a set of validated questionnaires at home, including questionnaires about medication use, quality of life, lifestyle, neuropsychological symptoms, autonomic symptoms, sleep, and vision, among others . These questionnaires are completed within 4weeks after each visit, via an online survey module.
Finally, a copy of the medical record of the participants primary care physician is requested. This provides a rich information source of known and unknown markers of disease progression.
Fig. 1
The Verily Study Watch , along with syncing/charging cradle and Study Hub. The photographs are owned by Verily and have a copyright. Verily kindly granted written permission to use and adopt if for this publication
Parkinson’s Disease Symptoms: Life Expectancy
Even though Parkinson’s disease is a serious, progressive condition, it is not considered a fatal illness. People who have Parkinson’s disease usually have the same average life expectancy as people without the disease.
But when the disease is in its advanced stages, Parkinson’s symptoms can lead to life-threatening complications, including:
- Falls that lead to fractured bones
- Pneumonia
- Choking
Thinking about the progression of Parkinson’s disease can be frightening. But proper treatments can help you live a full, productive life for years to come. And researchers hope to one day find ways to halt the progression of Parkinson’s and restore lost functioning.
Living With Parkinsons Disease
Depending on severity, life can look very different for a person coping with Parkinsons Disease. As a loved one, your top priority will be their comfort, peace of mind and safety. Dr. Shprecher offered some advice, regardless of the diseases progression. Besides movement issues Parkinsons Disease can cause a wide variety of symptoms including drooling, constipation, low blood pressure when standing up, voice problems, depression, anxiety, sleep problems, hallucinations and dementia.; Therefore, regular visits with a neurologist;experienced with Parkinsons are important to make sure the diagnosis is on target, and the symptoms are monitored and addressed.; Because changes in your other medications can affect your Parkinsons symptoms, you should remind each member of your healthcare team to send a copy of your clinic note after every appointment.
Dr. Shprecher also added that maintaining a healthy diet and getting regular exercise can help improve quality of life.;Physical and speech therapists;are welcome additions to any caregiving team.
Read Also: Is Parkinson’s Disease Fatal
Oxidative Stress And Parkinsons Disease
Neurodegeneration is a multifaceted process and the mechanisms that result in cellular death are linked to events that cause oxidative stress. Although there is much evidence in favor of this hypothesis; there is no definitive study. In the current section, the discussion focuses on: the markers of oxidative stress that have been observed in PD; how the cell becomes stressed; what oxidative damage does to cells; and cellular defense mechanisms to help overcome oxidative stress.
Oxidative stress is a key pathological process that is common to all neurodegenerative diseases. While oxidative stress occurs over the entire brain, the nigral environment appears to be more sensitive to oxidative stress . Although loss of DA producing neurons is seen in both normal aging individuals and PD cases it has been hypothesized that these catecholaminergic neurons are particularly susceptible to oxidative stress .
Superoxide as well as ascorbic acid and thiols can rereduce oxidized metals via the Haber-Weiss reaction to produce OH from H2O2. Purines and pyrmidines in DNA and RNA can be attacked by the hydroxyl radical, producing irreparable breakages and oxidized RNA products such as 8-OHG .
An Oxidative stress model suggesting the roles of Iron , -synuclein and Dopamine in the propagation of neurondegeneration in PD . The stars indicate the points on the pathways that MPAC compounds can influence the reaction.
2.4.1. Metals in PD
2.4.1.1. Iron
2.4.1.2. Iron Distribution in the Brain
Medicines For Parkinson’s Disease
Medicines prescribed for Parkinson’s include:
- Drugs that increase the level of dopamine in the brain
- Drugs that affect other brain chemicals in the body
- Drugs that help control nonmotor symptoms
The main therapy for Parkinson’s is levodopa, also called L-dopa. Nerve cells use levodopa to make dopamine to replenish the brain’s dwindling supply. Usually, people take levodopa along with another medication called carbidopa. Carbidopa prevents or reduces some of the side effects of levodopa therapysuch as nausea, vomiting, low blood pressure, and restlessnessand reduces the amount of levodopa needed to improve symptoms.
People with Parkinson’s should never stop taking levodopa without telling their doctor. Suddenly stopping the drug may have serious side effects, such as being unable to move or having difficulty breathing.
Other medicines used to treat Parkinsons symptoms include:
- Dopamine agonists to mimic the role of dopamine in the brain
- MAO-B inhibitors to slow down an enzyme that breaks down dopamine in the brain
- COMT inhibitors to help break down dopamine
- Amantadine, an old antiviral drug, to reduce involuntary movements
- Anticholinergic drugs to reduce tremors and muscle rigidity
Recommended Reading: Is Parkinson’s An Autoimmune Disease
Gait Progression Over 6 Years In Parkinsons Disease: Effects Of Age Medication And Pathology
- 1Translational and Clinical Research Institute, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne, United Kingdom
- 2The Newcastle upon Tyne NHS Foundation Trust, Newcastle upon Tyne, United Kingdom
- 3Auckland University of Technology, Auckland, New Zealand
- 4Department of Sport, Exercise and Rehabilitation, Northumbria University, Newcastle upon Tyne, United Kingdom
- 5Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne, United Kingdom
- 6School of Biomedical, Nutritional and Sport Sciences, Newcastle University, Newcastle upon Tyne, United Kingdom
Background: Gait disturbance is an early, cardinal feature of Parkinsons disease associated with falls and reduced physical activity. Progression of gait impairment in Parkinsons disease is not well characterized and a better understanding is imperative to mitigate impairment. Subtle gait impairments progress in early disease despite optimal dopaminergic medication. Evaluating gait disturbances over longer periods, accounting for typical aging and dopaminergic medication changes, will enable a better understanding of gait changes and inform targeted therapies for early disease. This study aimed to describe gait progression over the first 6 years of PD by delineating changes associated with aging, medication, and pathology.
Ophthalmic Assessments And Retinal Structure
A comprehensive ophthalmic assessment was performed by a consultant ophthalmologist and included slit-lamp examination and measurement of intra-ocular pressures.
Inner retinal layer structure was measured using high-resolution spectral-domain optical coherence tomography after pharmacological mydriasis according to a standard protocol , as described previously . Automatic layer segmentation was applied to compute the thickness of each retinal layer and manually corrected and verified as previously described . We focused on the ganglion cell layer and inner plexiform layer as these are the locations of dopaminergic amacrine cells, with most evidence for thinning in Parkinsons disease and we have previously shown an association between cognitive risk and these layers in Parkinsons disease .
You May Like: How To Take Mannitol For Parkinson’s
What Factors Are Predictive Of The Rate Of Progression Of Parkinson Disease
The American Academy of Neurology notes that the following clinical features may help predict the rate of progression of Parkinson disease :
-
Older age at onset and initial rigidity/hypokinesia can be used to predict a more rapid rate of motor progression in those with newly diagnosed Parkinson disease and earlier development of cognitive decline and dementia; however, initially presenting with tremor may predict a more benign disease course and longer therapeutic benefit from levodopa
-
A faster rate of motor progression may also be predicted if the patient is male, has associated comorbidities, and has postural instability/gait difficulty
-
Older age at onset, dementia, and decreased responsiveness to dopaminergic therapy may predict earlier nursing home placement and decreased survival
What You Can Do
As of 2021, there is no definite cure for Parkinsons disease. There is also no definite known cause. Its likely due to a combination of an individuals susceptibility and environmental factors. Most cases of Parkinsons disease happen without a genetic link.
According to research published in 2012, only report having a family member with the disease. Many toxins are suspected and have been studied, but no single substance can be reliably linked to Parkinsons.
However, research is ongoing. Its estimated that
Recommended Reading: What Does Parkinson’s Smell Like
Medications For The Disease Are Toxic
There are several medications available for Parkinsons disease, but the most commonly used is Sinemet . It is designed to restore levels of dopamine in the brain. The medication works well, but a myth that it was toxic began circulating and is still somehow commonly accepted. The truth is as long as the medicine is being used properly and the dose is where it should be, it is completely safe and can benefit people with Parkinson’s disease.
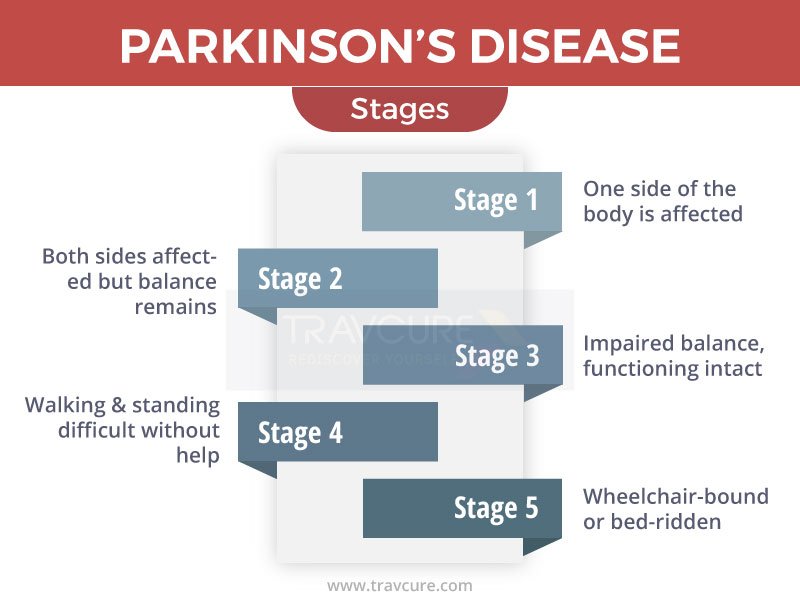
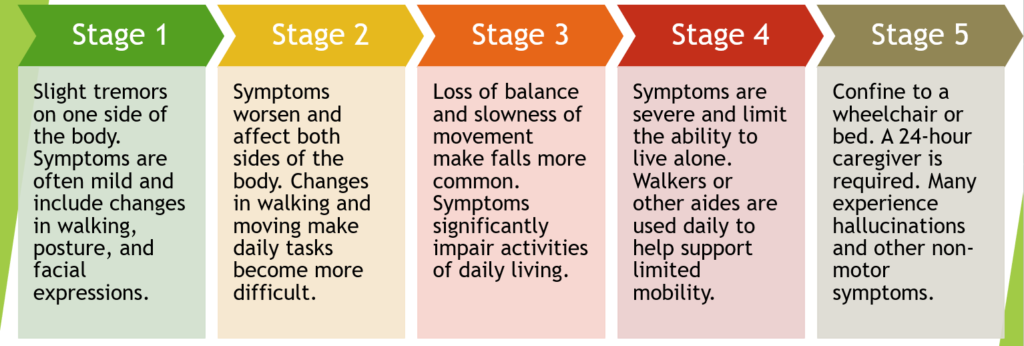
What Are The Stages Of Parkinson’s Disease
Parkinson’s disease is a progressive neurological disorder. There are five generally recognized stages of Parkinson’s disease. The progression of the disease may vary from one patient to the next, and not all patients will experience all five stages of Parkinson’s disease in their generally accepted order. Symptoms may also vary throughout the progression of Parkinson’s disease.
While doctors have currently identified five distinct stages of Parkinson’s disease, not all patients will experience a straightforward disease progression that passes through all five stages, one after the other. Nor will all patients remain in all stages for the same length of time. The five stages of Parkinson’s often vary in duration from patient to patient. Disease progression is generally considered impossible to predict, with some patients experiencing all stages while other patients skipping from an early stage to an advanced stage without passing through the stages in between.
Don’t Miss: Is Parkinson’s Disease Genetic
Stooping Or Hunching Over
Are you not standing up as straight as you used to? If you or your family or friends notice that you seem to be stooping, leaning or slouching when you stand, it could be a sign of Parkinson’s disease .
What is normal?If you have pain from an injury or if you are sick, it might cause you to stand crookedly. Also, a problem with your bones can make you hunch over.
How Can Hospice Help Your Loved One In The Final Stages Of Parkinsons Disease
Hospice care is an extra layer of support to help you care for your loved one with end-stage Parkinsons disease. It is a special kind of care that provides comfort, support, and dignity at the end of life.
The comprehensive program focuses on physical, emotional, and spiritual quality of life through the help of a team of experts. The team includes a board-certified physician, nurse, social worker, certified home health aide , spiritual support counselor, and volunteer.
The nurse will explain the prognosis and what to expect in the upcoming days or weeks. They will also monitor pain and other symptoms. The CHHA helps with personal care needs like bathing and changing bed linens. The social worker helps address social, emotional and practical challenges including complex and inter-related needs. The spiritual support counselor helps explore spiritual concerns.
Most importantly, the hospice team will be there for you during this difficult time, ;bringing you peace of mind. The team is on call 24 hours a day even at 2:00 am.
Hospice is about making your final months and weeks as good as possible. This means focusing on what really matters to you.
Don’t Miss: Can Adderall Cause Parkinson’s
Niacin Is A Protector With Metal Binding Properties
Vitamin B3, or niacin, also known as nicotinic acid, may alleviate certain types of early-onset PD symptoms . Niacin has been shown to attenuate neuroinflammation through an action on niacin receptor 1 , also known as GPR109A and may have a therapeutic potential toward PD . Although moderate amounts of niacin are found in a number of foods, including chicken, turkey, beef, peanut and mushrooms, the vitamin can be supplemented in therapeutic doses as tablets. In MPTP exposed rodents, the administration of nicotinamide gave a dose-dependent saving of striatal DA levels and SN neurons . Niacin, which is a precursor for nicotinamide adenine dinucleotide needed for DA production, may serve several purposes, i.e., reduce inflammation through NIARC1-related mechanisms, increase DA synthesis in the striatum through NADPH supply and increase NAD/NADH ratio to restore complex 1 functions in mitochondria. Niacin can also bind transition metal ions including Fe into stable complexes .
Caregiving In The Late Stages Of Parkinsons Disease
In late-stage PD, patients have significant mobility challenges. Caregivers likely need to provide more hands-on assistance to help them get around the house. Its important that caregivers learn safe and effective methods to provide help without injuring themselves. Physical therapists can be a great resource to assess an individual situation and teach effective ways of giving assistance.3
Freezing, a sudden but temporary inability to move, can become more common in late-stage PD. Freezing often happens when initiating movement or navigating around obstacles, and freezing episodes contribute to falls. Caregivers can help their loved one overcome freezing by providing a visual cue to step over, like a laser pointer, or using music or rhythm for the person with PD to walk to.3
Late stage PD can make daily activities, such as getting dressed, much more challenging. Caregivers can make getting dressed easier by ensuring adequate time to account for slow movement, choosing a time when medications are “on” and working well, and assembling all necessary items close to the person. Allowing the person with PD to do as much as they can gives them a sense of participation in the process.3
Don’t Miss: Parkinson’s Disease Autoimmune
What Causes Parkinson’s Disease
Parkinson’s disease occurs when nerve cells, or neurons, in an area of the brain that controls movement become impaired and/or die. Normally, these neurons produce an important brain chemical known as dopamine. When the neurons die or become impaired, they produce less dopamine, which causes the movement problems of Parkinson’s. Scientists still do not know what causes cells that produce dopamine to die.
People with Parkinson’s also lose the nerve endings that produce norepinephrine, the main chemical messenger of the sympathetic nervous system, which controls many functions of the body, such as heart rate and blood pressure. The loss of norepinephrine might help explain some of the non-movement features of Parkinson’s, such as fatigue, irregular blood pressure, decreased movement of food through the digestive tract, and sudden drop in blood pressure when a person stands up from a sitting or lying-down position.
Many brain cells of people with Parkinson’s contain Lewy bodies, unusual clumps of the protein alpha-synuclein. Scientists are trying to better understand the normal and abnormal functions of alpha-synuclein and its relationship to genetic mutations that impact Parkinsons disease and Lewy body dementia.
Mri Acquisition And Image Analysis
In the discovery cohort, high-resolution anatomical T1-weighted images were acquired at 3 T on a Siemens Prism-fit MRI system with a 64-channel head coil . Susceptibility-weighted MRI images were obtained from a 2 × 1-accelerated, 3D flow-compensated spoiled-gradient-recalled echo sequence. Flip angle 12°; echo time, 18 ms; repetition time, 25 ms; and receiver bandwidth, 110 Hz/pixel. Matrix size was 204 × 224 ×160 with 1 × 1 × 1 mm3 voxel resolution . Multishell diffusion weighted imaging was acquired with the following parameters: b=50 s/mm2 , b=300 s/mm2 , b=1000 s/mm2 , b=2000 s/mm2 ; 2 × 2 × 2 mm isotropic voxels, echo time = 3260 ms, repetition time = 58 ms, 72 slices, 2 mm thickness, acceleration factor = 2.
Because of MRI safety requirements, five patients and one control subject were unable to undergo MRI scanning. All images were assessed visually for quality, including artefacts such as motion and distortions. All T1-weighted scans were bias-corrected via the FreeSurfer protocol with parameters optimized for 3 T. Cortical reconstruction and volumetric segmentation of MRI scans for both cohorts was performed using FreeSurfer-v6.0 software . This involves skull stripping, volumetric labelling, intensity normalization, grey/white matter segmentation and registration to established surface atlases. Cortical thickness was calculated as the closest distance from the greywhite matter boundary to the grey matterCSF boundary at each vertex.
Also Check: Can You Die From Parkinson\’s Disease
Multivariate Models Of Baseline Predictors Of Long
Table;3 shows significant baseline predictors of change from baseline of total MDS-UPRDS score in the OFF state. These were baseline disease duration, MDS-UPDRS total score in the OFF state, male gender, CSF amyloid-142 , mean striatum SBR , orthostatic SBP , and SCOPA-AUT .
Table;3
Final results of mixed models examining baseline predictors of outcomes. Only variables associated with the outcome at a p-value of 0.10 are listed here. For the full model, see Supplementary Table 2. BMI, body mass index; CSF, cerebrospinal fluid; ESS, Epworth Sleepiness Scale; PIGD, postural instability gait disorder; MoCA, Montreal Cognitive Assessment; RBDSQ, REM Sleep Behavior Disorder Questionnaire; SBR, specific binding ratio; SCOPA-AUT, Scales for Outcomes in ParkinsonsAutonomic; SBP, systolic blood pressure
| Outcome |
| 0.3251 | 0.0786 |
iAge, gender, disease duration, and the baseline value of the outcome were forced into each model.
For the model examining change from baseline in total MDS-UPDRS score in the ON state as the outcome, significant predictors included baseline disease duration, baseline total MDS-UPDRS score in ON state,, baseline SCOPA-AUT, baseline CSF amyloid-142, male gender, 1-yr- in total MDS-UPDRS score in the ON state , and 1-yr- in SCOPA-AUT . Compared to the model only containing the baseline predictors, MoCA was not a significant predictor.
