Other Causes Of Parkinsonism
“Parkinsonism” is the umbrella term used to describe the symptoms of tremors, muscle rigidity and slowness of movement.
Parkinson’s disease is the most common type of parkinsonism, but there are also some rarer types where a specific cause can be identified.
These include parkinsonism caused by:
- medication where symptoms develop after taking certain medications, such as some types of antipsychotic medication, and usually improve once the medication is stopped
- other progressive brain conditions such as progressive supranuclear palsy, multiple systems atrophy and corticobasal degeneration
- cerebrovascular disease where a series of small strokes cause several parts of the brain to die
You can read more about parkinsonism on the Parkinson’s UK website.
Page last reviewed: 30 April 2019 Next review due: 30 April 2022
Genes Linked To Parkinsons Disease
Theres a long list of genes known to contribute to Parkinsons, and there may be many more yet to be discovered. Here are some of the main players:
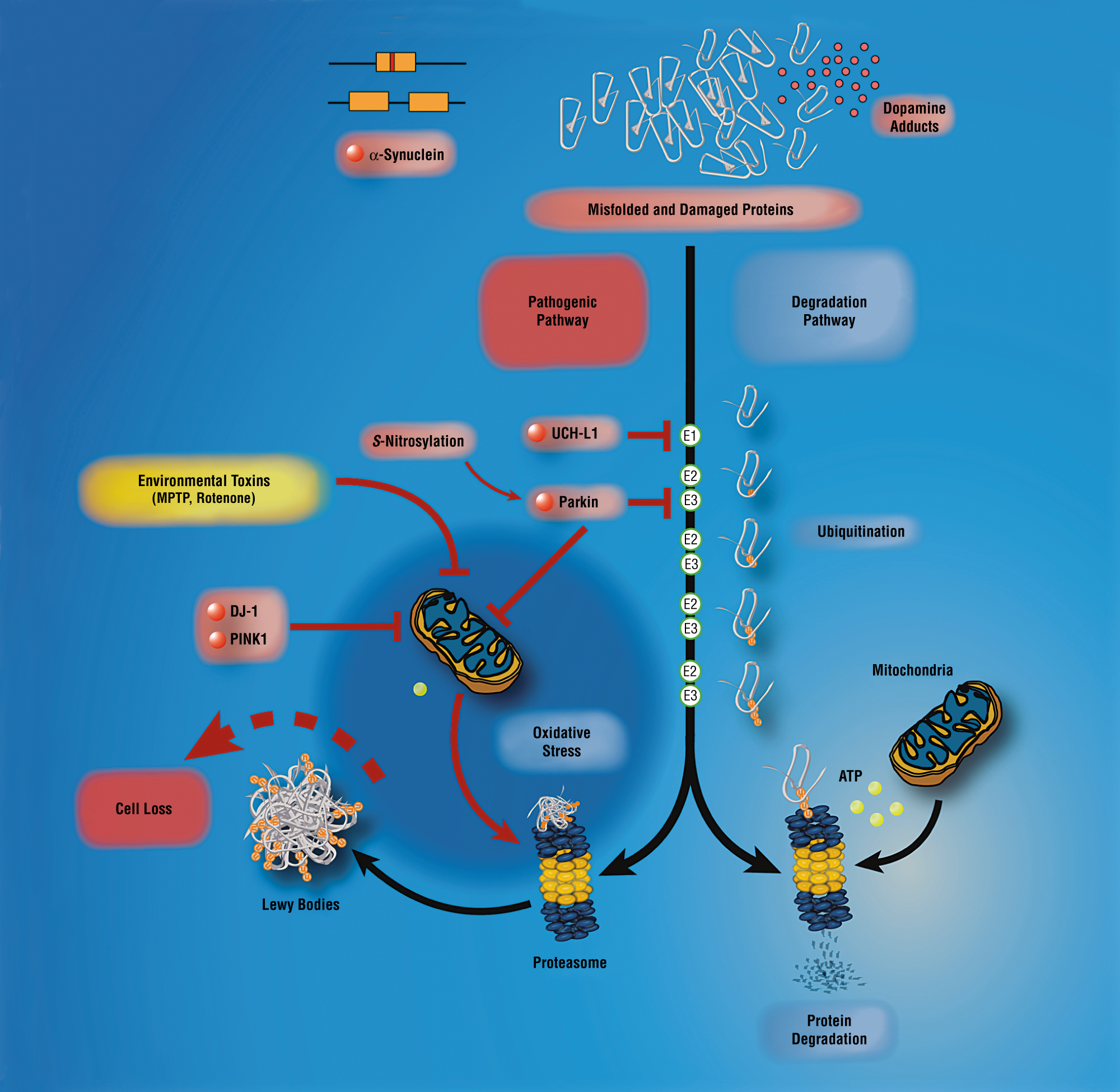
SNCA: SNCA makes the protein alpha-synuclein. In brain cells of individuals with Parkinsons disease, this protein gathers in clumps called Lewy bodies. Mutations in the SNCA gene occur in early-onset Parkinsons disease.
PARK2: The PARK2 gene makes the protein parkin, which normally helps cells break down and recycle proteins.
PARK7: Mutations in this gene cause a rare form of early-onset Parkinsons disease. The PARK7 gene makes the protein DJ-1, which protects against mitochondrial stress.
PINK1: The protein made by PINK1 is a protein kinase that protects mitochondria from stress. PINK1 mutations occur in early-onset Parkinsons disease.
LRRK2: The protein made by LRRK2 is also a protein kinase. Mutations in the LRRK2 gene have been linked to late-onset Parkinsons disease.
Among inherited cases of Parkinsons, the inheritance patterns differ depending on the genes involved. If the LRRK2 or SNCA genes are involved, Parkinsons is likely inherited from just one parent. Thats called an autosomal dominant pattern, which is when you only need one copy of a gene to be altered for the disorder to happen.
Assessment Of Family History
The majority of our identified studies use self reporting or self administered questionnaires to assess family history of PD. Categorising PD cases who have relatives with isolated tremor as having a positive family history, can significantly increase the number of familial cases, especially among early onset PD cases.
Performing individual examinations may increase the precision with which a diagnosis of PD is made in relatives of cases and controls, rather than reliance on patient reporting of diagnoses or symptoms such as tremor. It has also been shown that significant numbers of previously unrecognised PD patients can be identified by examination despite a negative family history. It can often however be difficult verifying familial diagnoses in diseases affecting the elderly as relatives are often deceased and not subjected to postmortem examination. Subclinical Parkinson’s disease, diagnosed on the basis of Lewy body pathology in people without prior symptoms of PD, is observed in up to 10% of individuals subjected to postmortem neuropathological examination. No study includes pathological examination of all relatives of both cases and controls, which currently represents the gold standard in diagnosing PD.
Also Check: Is Parkinsons Disease Fatal
Epigenomic Alterations Linked With Pd In Patient
The ability to capture unique epigenomic alterations associated with PD remains an important challenge. Reprogramming fibroblasts to iPSCs may erase age-associated and naive epigenetic signatures which could contribute to sporadic PD pathophysiology. However, an epigenetic phenotype was reported in iPSC-derived PD patient neurons,. Neuronal lines derived from LRRK2 and sporadic patients exhibited epigenomic alterations when compared with healthy controls. Hypermethylation was prominent in gene regulatory regions associated with the downregulation of transcription factors FOXA1, NR3C1, HNF4A, and FOSL2. Interestingly, LRRK2 mutant and sporadic PD patient neurons shared similar methylation patterns, which were absent in the original donor fibroblasts. A spontaneous increase in the number of DNA strand breaks and genomic damage in PD patient-derived neurons could indirectly impact genomic regulation.
What Determines Who Gets Parkinson’s Disease
In most cases inheriting a non-working copy of a single gene will not cause someone to develop Parkinson’s disease. We believe that many other complicating factors such as additional genes and environmental factors determine who will get the condition, when they get it and how it affects them. In the families we have studied, some people who inherit the gene develop the condition and others live their entire lives without showing any symptoms. There is a lot of research on genes and the environment that is attempting to understand how all these factors interact.
Genetic Testing in Parkinson’s Disease
Genetic testing has recently become available for the parkin and PINK1 genes. Parkin is a large gene and testing is difficult. At the current stage of understanding, testing is likely to give a meaningful result only for people who develop the condition before the age of 30 years. PINK1 appears to be a rare cause of inherited Parkinson’s disease. A small percentage of those developing the condition at an early age appear to carry mutations in the PINK1 gene. Genetic testing for the PARK7, SNCA and LRRK2 genes is also available.
Additional Resources
Don’t Miss: Can Parkinson’s Run In The Family
How Do I Prevent Falls From Common Hazards
- Floors: Remove all loose wires, cords, and throw rugs. Minimize clutter. Make sure rugs are anchored and smooth. Keep furniture in its usual place.
- Bathroom: Install grab bars and non-skid tape in the tub or shower. Use non-skid bath mats on the floor or install wall-to-wall carpeting.
- Lighting: Make sure halls, stairways, and entrances are well-lit. Install a night light in your bathroom or hallway and staircase. Turn lights on if you get up in the middle of the night. Make sure lamps or light switches are within reach of the bed if you have to get up during the night.
- Kitchen: Install non-skid rubber mats near the sink and stove. Clean spills immediately.
- Stairs: Make sure treads, rails, and rugs are secure. Install a rail on both sides of the stairs. If stairs are a threat, it might be helpful to arrange most of your activities on the lower level to reduce the number of times you must climb the stairs.
- Entrances and doorways: Install metal handles on the walls adjacent to the doorknobs of all doors to make it more secure as you travel through the doorway.
Identification Of New Genes And Risk Factors For Pd
New PD-linked genes or PD risk factors can be identified by gene mapping or candidate gene approaches. Gene mapping in human diseases is the localization of genes underlying the clinical phenotypes of the disease on the basis of correlation with DNA variants , without the need for prior hypotheses about biological function. Genetic mapping methods include linkage analysis and genome-wide association studies. Alternatively, based on their known function, levels of expression, or mode of interaction , some genes can be considered plausible candidates, and as such, tested for in cohorts of patients.
Also Check: What Is The Life Expectancy Of Someone With Parkinson’s Disease
Neuroinflammation Exacerbates Neurodegeneration In Sporadic Pd
Midbrain neurons derived from sporadic patients showed increased susceptibility to the effects of adaptive immune cells. Sporadic patient neuronal lines co-cultured with T-lymphocytes exhibited substantial signs of cell death mediated by IL-17IL-17R signaling and activation of NFkB. Similarly, IL-17 treatment resulted in increased neuronal death. Inflammation in the central nervous system and periphery are key hallmarks of PD. Increasing evidence implicates the role of microglia in neuronal loss, though the underlying mechanisms remain to be determined,. RNA-seq analysis of astrocytes derived from LRRK2-G2019S iPSCs highlighted dysregulation in genes involved in the extracellular matrix, which may reduce the neuroprotective capacity of astrocytes in PD. Investigating the role of neuroinflammation in patient-derived microglia may also contribute to the understanding of the selective vulnerability of mDA neurons in sporadic and late-onset PD.
Diagnostic Features Of Pd
The cardinal pathologic feature of PD is the loss of dopaminergic neurons in the substantia nigra with intracytoplasmic inclusions in the remaining, intact nigral neurons., Symptoms of PD typically onset when 5080% of the dopaminergic neurons in the substantia nigra are no longer functioning. Traditionally, the presence of Lewy bodies was required for pathologic confirmation of PD however, it has been recognized recently that nigral pathology may occur in the absence of Lewy bodies.
Several other neurologic entities must be considered as part of the differential diagnosis of PD. They include parkinsonism-predominant multiple system atrophy , progressive supranuclear palsy, corticobasal degeneration, essential tremor, drug-induced parkinsonism, postencephalitic conditions, Lewy body dementia, and AD. Parkinsonism can also be a prominent feature of some Mendelian disorders, including spinocerebellar ataxias , Huntington disease, dopa-responsive dystonia, familial prion disease, frontotemporal dementia with parkinsonism-17, Wilson disease, and X-linked dystonia-parkinsonism syndrome . Laboratory or radiologic studies cannot be used to confirm PD however, they are useful in excluding alternative diagnoses, such as stroke, tumor, and thyroid disease.
Also Check: What Is The Life Expectancy Of Someone With Parkinson’s Disease
Thanks For Signing Up
We are proud to have you as a part of our community. To ensure you receive the latest Parkinsons news, research updates and more, please check your email for a message from us. If you do not see our email, it may be in your spam folder. Just mark as not spam and you should receive our emails as expected.
Genetic Study Of Lewy Body Dementia Supports Ties To Alzheimers And Parkinsons Diseases
NIH-led study locates five genes that may play a critical role in Lewy body dementia.
In a study led by National Institutes of Health researchers, scientists found that five genes may play a critical role in determining whether a person will suffer from Lewy body dementia, a devastating disorder that riddles the brain with clumps of abnormal protein deposits called Lewy bodies. Lewy bodies are also a hallmark of Parkinsons disease. The results, published in Nature Genetics, not only supported the diseases ties to Parkinsons disease but also suggested that people who have Lewy body dementia may share similar genetic profiles to those who have Alzheimers disease.
Lewy body dementia is a devastating brain disorder for which we have no effective treatments. Patients often appear to suffer the worst of both Alzheimers and Parkinsons diseases. Our results support the idea that this may be because Lewy body dementia is caused by a spectrum of problems that can be seen in both disorders, said Sonja Scholz, M.D., Ph.D., investigator at the NIHs National Institute of Neurological Disorders and Stroke and the senior author of the study. We hope that these results will act as a blueprint for understanding the disease and developing new treatments.
The study was led by Dr. Scholzs team and researchers in the lab of Bryan J. Traynor, M.D., Ph.D., senior investigator at the NIHs National Institute on Aging .
Article:
NIHTurning Discovery Into Health®
Also Check: Weighted Bracelet For Essential Tremors
Tips To Slow Or Stop Progression
Some suggest that you may be able to delay some of the effects of Parkinsons disease through regular physical activity. Ideally this would include a combination of exercise that includes:
- aerobic activity
- balance training
- functional activities
The NINDS has funded a number of studies to learn more about the impact of exercise, including whether exercise might help people delay the need for medication.
Genetic Risk For Parkinson’s Disease
If you have a genetic mutation associated with Parkinson’s, will you get the disease? Not necessarily. Some mutations carry a greater risk, but none bring a 100 percent chance of developing Parkinson’s disease. There are many Parkinson’s risk genes where a mutation means a very small increased likelihood of Parkinson’s. Researchers are looking for other factors that either push or protect someone with a gene mutation to or from having Parkinson’s. Your doctor and/or a genetic counselor can discuss the risk associated with different Parkinson’s genes and what your results may mean for you and your loved ones.
Recommended Reading: Is Parkinson’s Disease Hereditary Or Genetic
Understanding The Lrrk2 Mutation
People withLRRK2 Parkinsonâs disease
Most people withLRRK2 Parkinsonâs disease have one normal gene and one mutated gene. This means they have both normal LRRK2 and an overactive version. The overactive LRRK2 causes certain cells in the brain to degenerate, resulting in the development of Parkinsonâs disease.
ESCAPE Bioâs approach
ESCAPE is developing an investigational therapy that only blocks the overactive LRRK2. The therapy is intended to be taken by mouth in pill or capsule form.
Studies Of Pd With Patient
The discovery of iPSC technology,, has offered the capacity to generate live brain tissue from healthy subjects and patients for studying neurodegenerative diseases. Directed reprogramming and neuronal differentiation of iPSCs allows the study of specific neuronal subtypes. Human-derived neurons offer a unique opportunity for modeling real cases of human genetic diseases in vitro. The ability to generate neurons both from PD patients and healthy control individuals allows the identification of early disease-linked phenotypes and provides a new paradigm for preclinical drug development and validation .
Fig. 3: Using brain cells generated from patient-derived iPSC to study PD in vitro.
You May Like: Parkinson’s Donations In Memory Of
How Often Does Parkinsons Run In The Family
Most Parkinsons cases have no connection to a genetic cause, but scientists have found that some gene mutations can heighten an individuals risk. Researchers believe that a better understanding of these genes may improve ways of identifying and treating the illness.
The National Institute of Neurological Disorders and Stroke reports that an estimated 15 to 25 percent of people with Parkinsons have a family history of the disorder. The Michael J. Fox Foundation for Parkinsons Research estimates that about 10 percent of cases are linked with a genetic cause.
Parkinsons doesnt stand out as a hereditary disease over and above any other chronic diseases that people deal with, says Rebecca Gilbert, MD, PhD, chief scientific officer for the American Parkinson Disease Association in New York City. But if you have a parent with Parkinsons disease, you have about a fourfold greater risk over the general population.
Still, that risk is relatively small. About 1 percent of the population over 60 has Parkinsons, according to the Michael J. Fox Foundation, and that number rises to about 4 percent for those who have a mother or father with the illness, according to Dr. Gilbert. The overall message is: Just because you have a gene linked to Parkinsons does not mean you will get the disease.
Can Parkinsons Be Passed From Parent To Child
Its rare for Parkinsons disease to be passed down from parent to child. Most cases of Parkinsons arent hereditary. But people who get early-onset Parkinsons disease are more likely to have inherited it.
Having a family history of Parkinsons disease may increase the risk that youll get it. This means that having a parent or sibling with Parkinsons slightly increases the risk.
In most cases, the cause of Parkinsons disease remains unknown. But researchers have identified multiple risk factors that can increase your chances of getting this disease.
Risk factors for Parkinsons disease include:
- mutations in specific genes associated with Parkinsons
- having a family history of Parkinsons or a first-degree family member with Parkinsons
- being older, especially above the age of 60
- exposure to herbicides and pesticides
- being assigned male at birth
- history of brain injury
Read Also: What Color Is The Ribbon For Parkinson’s
Environmental And Genetic Factors
Scientists are also working to learn more about environmental factors and genetic factors that might contribute to the risk of developing Parkinsons. One recent genetic research breakthrough is the development of a DNA chip called NeuroX, which could potentially determine a persons risk, but more research is needed.
Parkinsons disease is the result of complicated combination of interconnected events, as described it. Since aging is the most common risk factor, future treatments may need to take degeneration of certain neurons into account.
When Should Genetic Testing Be Done
Your physician may suggest it if your Parkinsons diagnosis comes at a young age , if multiple relatives in your family history have also been diagnosed with the same, or if you are at high risk for familial Parkinsons based on your ethnicity .
However, what is the benefit of having the testing done at the present time? The information may be important for family planning for some individuals although as I said even if the gene is passed on it does not necessarily equal development of the disease. The risk, though, is higher in dominant genetic mutations versus recessive ones, on average if a person has a first-degree relative with Parkinsons their risk of developing the disease is 4 to 9 percent more than the general population.
Keep in mind that currently for the person being tested, there is no change in the treatment of their Parkinsons disease based on genetic findings. In the future, however, when there are treatments to slow down the onset of the disease or to prevent it from developing at all, then the identification of those people at risk will be very important.
Read Also: What Color Is The Ribbon For Parkinson’s
Mjff Fellow Working To Raise Black Minority Presence In Research
Parkinsons affects over six million people worldwide, including almost one million in the U.S. Because disease knowledge has come from relatively homogenous populations of patients, evidence is lacking on its incidence, morbidity, or mortality across a variety of socioeconomic or ethnic groups.
Poor representation in research leads to an incomplete picture of Parkinsons. Evidence suggests that, across different ethnic groups, disease symptoms present differently. Genetic variation and other factors specific to these patients also impact therapy effectiveness and how medications are metabolized.
In the minority of PD clinical trials reporting race and ethnicity data, only 8% of participants were non-white, the investigators wrote.
To reduce health inequities and advance treatments for all Parkinsons patients, the MJFF described four areas of action: identifying barriers and delivering solutions to research participation, funding research that includes greater diversity, building a workforce of doctors and researchers committed to equity, and supporting a more holistic understanding of Parkinsons.
While MJFF efforts in these areas are currently not sufficient to fully reduce health disparities in PD, our hope is that they will serve as solid starting points from which to learn and iterate expanded efforts, the MJFF team wrote.
