How You Lose Dopamine Production
Damaged nerve cells can be what results in a decreased ability of the brain to create dopamine. Generally, some kind of degradation of the brain cells will reduce dopamine production.
There is some genetic link to parkinsons disease. For example, specific genetic mutations can impact the dopamine production. Far more common is that exposure to something in the environment can impact the brain.
Genes Connected To Parkinson’s
SNCA
In 1997, researchers at the National Institutes of Health made the first Parkinsons genetic connection, discovering that mutations in the SNCA gene were common in several families who had many members with Parkinsons. While mutations in this gene are rare, they have taught us invaluable information about PD.
The SNCA gene instructs the body to make the protein alpha-synuclein, which for reasons that are not yet known, clumps in the brain cells of people with PD. Study of the SNCA gene led to this understanding that applies to nearly all people with the disease. Since then, alpha-synuclein has been a major target of new drugs for Parkinsons.
LRRK2
Another gene that plays a role in Parkinsons is LRRK2, which accounts for one to two percent of all cases. For people of particular ethnic backgrounds Ashkenazi Jewish and North African Berber descent mutations in the LRRK2 gene account for a much greater number of cases than in the general PD population. Mutations in LRRK2 lead to too much activity of the LRRK2 protein, though recent research indicates people without a mutation can also have overactivity of LRRK2.
Shake It Up are funding several studies into LRRK2 and you can learn more about these here.GBA
PRKN
Assessment Of Family History
The majority of our identified studies use self reporting or self administered questionnaires to assess family history of PD. Categorising PD cases who have relatives with isolated tremor as having a positive family history, can significantly increase the number of familial cases, especially among early onset PD cases.
Performing individual examinations may increase the precision with which a diagnosis of PD is made in relatives of cases and controls, rather than reliance on patient reporting of diagnoses or symptoms such as tremor. It has also been shown that significant numbers of previously unrecognised PD patients can be identified by examination despite a negative family history. It can often however be difficult verifying familial diagnoses in diseases affecting the elderly as relatives are often deceased and not subjected to postmortem examination. Subclinical Parkinson’s disease, diagnosed on the basis of Lewy body pathology in people without prior symptoms of PD, is observed in up to 10% of individuals subjected to postmortem neuropathological examination. No study includes pathological examination of all relatives of both cases and controls, which currently represents the gold standard in diagnosing PD.
Recommended Reading: Stages Of Parkinson Disease Life Expectancy
Thanks For Signing Up
We are proud to have you as a part of our community. To ensure you receive the latest Parkinsons news, research updates and more, please check your email for a message from us. If you do not see our email, it may be in your spam folder. Just mark as not spam and you should receive our emails as expected.
What If I Carry The Gene

There are ongoing clinical trials testing therapies to treat people who have Parkinson’s and carry certain gene mutations. Proving that it can be important to know which gene mutation you carry. Consult with your doctor when considering a genetic test to determine if you are eligible to participate in gene-based clinical trials.
The Parkinsons Foundation study, PD GENEration: Mapping the Future of Parkinsons Disease, is the first national study to offer genetic testing and counseling at no cost for those with a confirmed Parkinsons diagnosis. Learn more and enroll at Parkinson.org/PDGENEration.
*Please note that not all content is available in both languages. If you are interested in receiving Spanish communications, we recommend selecting both” to stay best informed on the Foundation’s work and the latest in PD news.
Recommended Reading: Life Expectancy For Parkinson’s Disease
Concluding Remarks And Future
Genes implicated in Mendelian forms of PD have provided new insights into the pathogenesis of the disease. The molecular pathways identified in monogenic cases may also be implicated in sporadic PD. The effect of dosage of SNCA on the phenotype of patients with duplications or triplications is illustrative. In addition, non-coding variants in this gene, thought to affect the level of expression in neurons, are associated with risk of the disease. The molecular mechanisms that contribute to PD and related disorders result in the death of dopaminergic neurons in vulnerable brain regions, and consequently the shared phenotype. However, known PD-causing genes account for only a small fraction of monogenic forms. Robust high-density SNP genotyping technologies and data analysis programs, combined with the analysis of copy number variations and large pathogenic genomic rearrangements, will identify novel loci. The clinical heterogeneity of parkinsonism is probably the cumulative effect of different gene-environment and/or genegene interactions. To identify risk variants in PD, association study methodology must be improved. Studies in isolated and heterogeneous populations, and approaches that minimize population stratification, are needed. Large-scale studies and publicly available GWA databases, crucial for statistical power, require collaborative efforts with shared sets of stringent clinical, genetic and analytic methods.
How Environmental Factors And Aging Can Be Recapitulated In Vitro
An obvious limitation of in vitro models is the lack of environmental context. The influence of nongenetic factors is not recapitulated in the basal phenotype of patient-derived neurons. For example, the influence of head trauma of a boxer with sporadic PD will not be recapitulated by default in reprogrammed neurons. An alternative would be to transplant the patient-derived neurons in animals and simulate the trauma on the animal. Similarly, influence of decades of aging of the human brain is difficult to reproduce in vitro in a few months within the boundaries of feasible experimental design. Brains in a dish will always be an imperfect experimental model. However, many tricks can be used to recapitulate the environmental and aging stress in vitro. Table summarizes a list of reagents that have already been used in iPSC neuronal culture to mimic oxidative stress, proteostatic stress, mitochondrial stress, synaptic stress, ER stress, inflammation, and cellular aging. An interesting example is progerin, a truncated form of lamin A associated with premature aging. Increasing the expression of progerin in iPSC neurons can recapitulate at least some aspect of cellular aging in vitro. Human iPSC-derived dopamine neurons overexpressing progerin displayed specific phenotypes such as neuromelanin accumulation. In addition, PD patient-derived neurons revealed disease-related phenotypes that required both genetic susceptibility and induced-aging in vitro.
Read Also: Rso For Parkinson\’s
What Else Do We Know
As scientists try to learn what’s at the root of Parkinson’s, they’re looking far and wide to pick up clues where they can.
They’ve found that people with Parkinson’s tend to have something called Lewy bodies in their brain. These are unusual clumps of a protein called alpha-synuclein. The protein itself is normal, but the clumps are not. And they’re found in parts of the brain that affect sleep and sense of smell, which could explain some symptoms of Parkinson’s not related to movement.
Your gut may also have a part in it, as some of its cells make dopamine, too. Some doctors think that this might be where the earliest signs of Parkinson’s show up, but that idea needs more research.
Reasons Why Parkinsons Disease Occurs
The scientific reason given for Parkinsons disease is that the patient has lost nerve cells in the part of the brain called the substantia nigra. A very important chemical called dopamine is produced by the substantia nigra. The loss of the ability to produce dopamine contributes to the early stages of Parkinsons disease.
Recommended Reading: Parkinson’s Mortality
Research Into Genes And Parkinsons
Our major effort now is understanding how mutations in these genes cause Parkinsons disease, says Dawson. SNCA, the gene responsible for making the protein that clumps in the brain and triggers symptoms, is particularly interesting.
Our research is trying to understand how alpha-synuclein works, how it travels through the brain, says Dawson. The latest theory is that it transfers from cell to cell, and our work supports that idea. Weve identified a protein that lets clumps of alpha-synuclein into cells, and we hope a therapy can be developed that interferes with that process.
Targeting Parkinsons-Linked Protein Could Neutralize 2 of the Diseases Causes
Researchers report they have discovered how two problem proteins known to cause Parkinsons disease are chemically linked, suggesting that someday, both could be neutralized by a single drug designed to target the link.
Predicting Risk Progression And Defining Etiological Subtypes Of Disease
Individually GWA identified loci confer relatively small amounts of disease risk however, the use of polygenic risk scores affords the ability to attribute a total known genetic risk score to an individual by summing their collective genetic risk. To date, the PRS reveals that, collectively, the 90 susceptibility loci confer considerable risk for disease, with those in the top decile of genetic risk being 6-fold more likely to have PD than those in the lowest decile of genetic risk . Additionally, by creating a composite risk score for PD diagnosis that combines the cumulative effect of genetic risk variants as well as the presence or absence of anosmia, age, sex, and family history, the ability to predict individuals at high risk for PD is remarkable, showing an AUC sensitivity of ~ 83.4% and specificity of ~ 90% .
Research focused on age at onset disease modifiers is one area where consistent effort has been seen. The largest PD age at onset GWAS to date included data from > 25K cases and identified two GWAS significant signals one at SNCA and the other was a protein-coding variant in TMEM175, both of which are known PD risk loci. Notably, these results showed that not all PD risk loci influence age at onset and therefore suggest the idea that risk and onset might operate through mechanisms that do not completely overlap.
Also Check: What Color Is The Ribbon For Parkinson’s
Omics Analysis Of Patient
To date 10/67 iPSC-PD studies analyzed have used proteomic, transcriptomic, or epigenomic profiling to phenotype PD patient-derived neurons,,,,,,,,. Omics analyses may be less biased and data-driven as opposed to purely hypothesis-driven. Data from omics studies can also help to describe biological relationships between complex intertwined cellular pathways and identify relevant druggable molecular pathways.
Parkinson’s Disease Genetic Influence
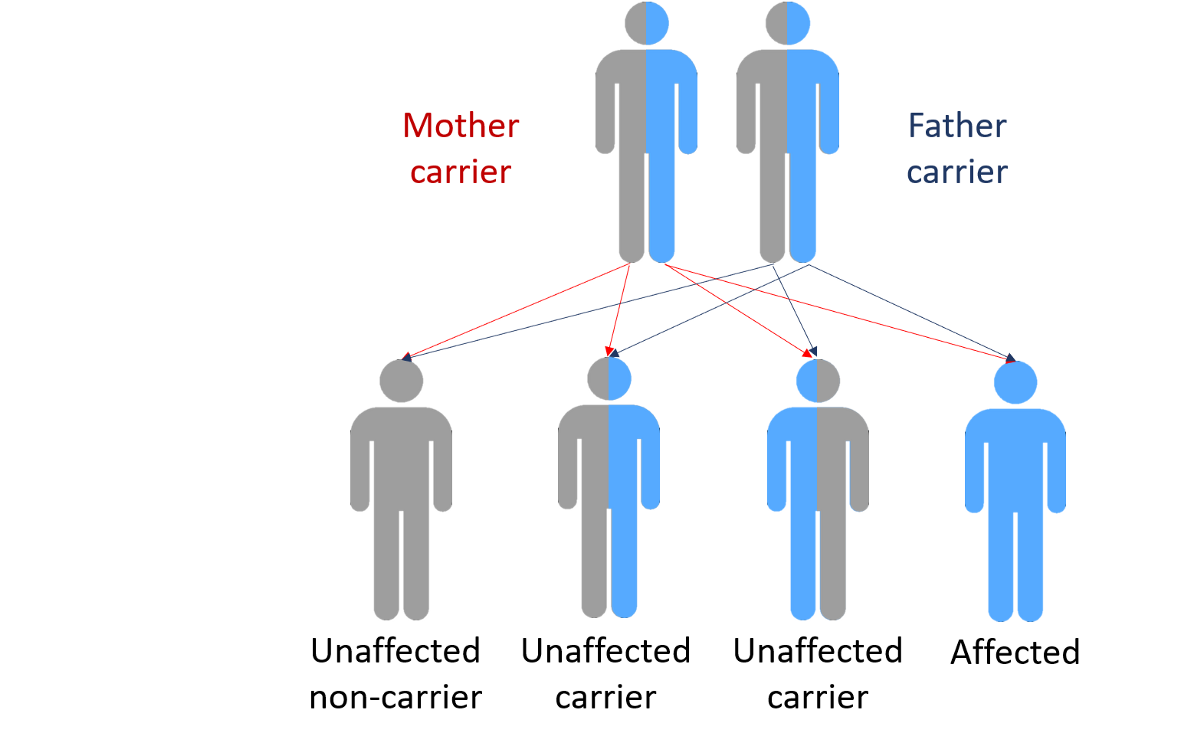
Recent developments in research gene research has found that genetic influence plays a large role in Parkinson’s disease. Five main genes that are believed to contribute to the disease have been identified and located. These include alpha-synuclein, Parkin, Ubiquitin carboxyl-terminal hydrolase, DJ-1 and SCA2. It has been found that mutations of these genes are some of the underlying causes. In some causes there can be multiple mutations in one gene. The effects of some of these mutations are now understood.
Researchers suspect that genes associated with the late onset of Parkinson’s Disease are susceptibility genes rather than causal genes. It is thought that environmental factors act on these gene, consequently leading to Parkinson’s disease. But the mechanism in which they do so is not yet known. Researchers believe that if they can work out this mechanism, they can disrupt it in some way, and therefore halt the onset of the disease.
The general consensus among researchers is that both genetic influence and environmental factors lead to the onset of Parkinson’s Disease. The mechanisms of the genetic influence of Parkinson’s Disease are still to be understood and much more research is required.
The content of this web site is intended to convey general educational information and should not be relied upon as a substitute for professional healthcare advice. More information.
Read Also: Gabapentin And Parkinson’s
Genetic Testing May Lead To A Cure
Although genetic testing can leave individuals with many unanswered questions, the data provided may further the study of the disease.
The more individuals you can work, the more things you can discover, says Cannon. We are interested in studying people who have a risk gene because the sooner we can learn how to stop it , the better off people will be.
Clinical trials are in progress to test therapies that target gene mutations, in particular GBA and LRRK2. Pharmaceutical companies conducting these studies need patients who test positive for specific gene variations. By getting tested, individuals have a chance to participate in research programs that may lead to a cure.
Gilbert points out that drugs that target specific mutations may benefit a larger group of Parkinsons patients.
The biochemical problem that happens when a person has an LRRK2 mutation might appear in someone else without an LRRK2 mutation but by another means, she says. So they may also benefit from medication developed for people with an LRRK2 mutation.
If you are interested in participating in a trial, the Michael J. Fox Foundation offers a roundup of the latest investigations currently being conducted and how to get involved.
Neuroinflammation Exacerbates Neurodegeneration In Sporadic Pd
Midbrain neurons derived from sporadic patients showed increased susceptibility to the effects of adaptive immune cells. Sporadic patient neuronal lines co-cultured with T-lymphocytes exhibited substantial signs of cell death mediated by IL-17IL-17R signaling and activation of NFkB. Similarly, IL-17 treatment resulted in increased neuronal death. Inflammation in the central nervous system and periphery are key hallmarks of PD. Increasing evidence implicates the role of microglia in neuronal loss, though the underlying mechanisms remain to be determined,. RNA-seq analysis of astrocytes derived from LRRK2-G2019S iPSCs highlighted dysregulation in genes involved in the extracellular matrix, which may reduce the neuroprotective capacity of astrocytes in PD. Investigating the role of neuroinflammation in patient-derived microglia may also contribute to the understanding of the selective vulnerability of mDA neurons in sporadic and late-onset PD.
You May Like: Life Expectancy Parkinsons
How Does Environment Come Into It
Your environment is a hard one to pin down. Partly, that’s because it covers a lot of ground. It’s everything that’s not your genes, which could mean where you live, what you eat, chemicals you’ve come into contact with, and more.
Not only that, but it could take years for the effects from something in your environment to show up. So far, doctors have a lot of clues but no smoking gun. So you could have people who live or work in an area around chemicals tied to Parkinson’s, but many of them don’t get it.
Some research shows links between Parkinson’s and:
- Agent Orange, a chemical used to destroy trees and crops in the Vietnam War.
- Certain chemicals used in farming, such as insecticides, herbicides, and fungicides.
- Some metals and chemicals used in factories, such as manganese, lead, and trichlorethylene .
These can come into play based on where you live, what you do for work, or if you served in the military. Sometimes, these chemicals seep into well water, so that’s one more way they can affect you.
Are Genes Responsible For Monogenic Disorders Also Susceptibility Factors
Associations detected by screening candidate genes in controls and patients cannot always be replicated in follow-up studies, and few candidate genes were confirmed in meta-analysis, because of potential biases and confounding factors, including population stratification, small sample size, misclassification and/or inappropriate statistical methods. Polymorphic variants in SNCA and LRRK2 genes, and heterozygous mutations in the GBA gene, however, have been validated as genetic susceptibility factors .
Nucleotide polymorphisms located close to the promoter region and throughout SNCA have been associated with sporadic PD, although much of the data is equivocal . Rep1 , a mixed nucleotide repeat, 10 kb upstream of the translational start of SNCA , has been confirmed as a risk factor , and synergy between an SNCA variant and a polymorphism in microtubule-associated protein tau , each of which increases the risk for the development of PD, has been detected . The combination of risk genotypes in SNCA and MAPT doubles the risk of PD, further supporting the notion that the related pathways contribute to neurodegenerative diseases . The risk associated with Rep1 does not interact, however, with herbicide exposure, an independent risk factor in PD .
Also Check: What Is The Life Expectancy Of Someone With Parkinson’s Disease
How Environmental Factors Could Cause Parkinsons Disease
Scientists differ about the extent that brain cells are impacted by environmental factors. However, the statistics associated with the disease show that the environment can play a very large role in whether parkinsons disease develops.
Most often, it is exposure to toxic chemicals that could play a role in the development of Parkinsons disease. Usually, these combine with genetic factors to produce the conditions that cause Parkinsons.
Increasing scientific evidence suggests that Parkinsons may be caused by environmental factors such as exposure to herbicides such as Paraquat.
Inferring Causal Relationships Across Phenotypic Traits And Exploring Shared Polygenic Risk
A corollary of abundant GWA data is the ability to test other traits that can predispose or protect individuals to disease. Access to large amounts of GWA data affords the opportunity to explore causal inferences between myriad traits and PD through Mendelian Randomization , the gold standard approach for causality in genetic studies that sits at the interface between observational epidemiology and interventional trials . MR has emerged as a statistical approach that uses genetic data in the form of SNPs to study whether an exposure exerts a causal effect in an outcome. This addresses the question of whether an observational association between a risk or protective factor and a disease of interest is consistent with a causal effect by focusing usually only on genome-wide significant SNPs. Put simply, genetic variants that explain variation in a certain exposure can be used as proxies to determine how a change in that exposure might influence a disease outcome. One of the key strengths of this method is that it relies on genetic variants that remain constant over the lifespan of an individual, are randomized during gametogenesis and fixed at conception, which means that genetic variants are not associated with all the confounder factors that affect an observational study.
In the PD field, two-sample MR has been applied considering both targeted exploration of causal associations, as well as hypothesis-generating approaches in a large-scale, high-throughput, unbiased manner.
Also Check: What State Has Highest Rate Of Parkinson’s
